
Ocean Microplastics Monitor
Client
Various
Practice Areas
Core Disciplines
Overview
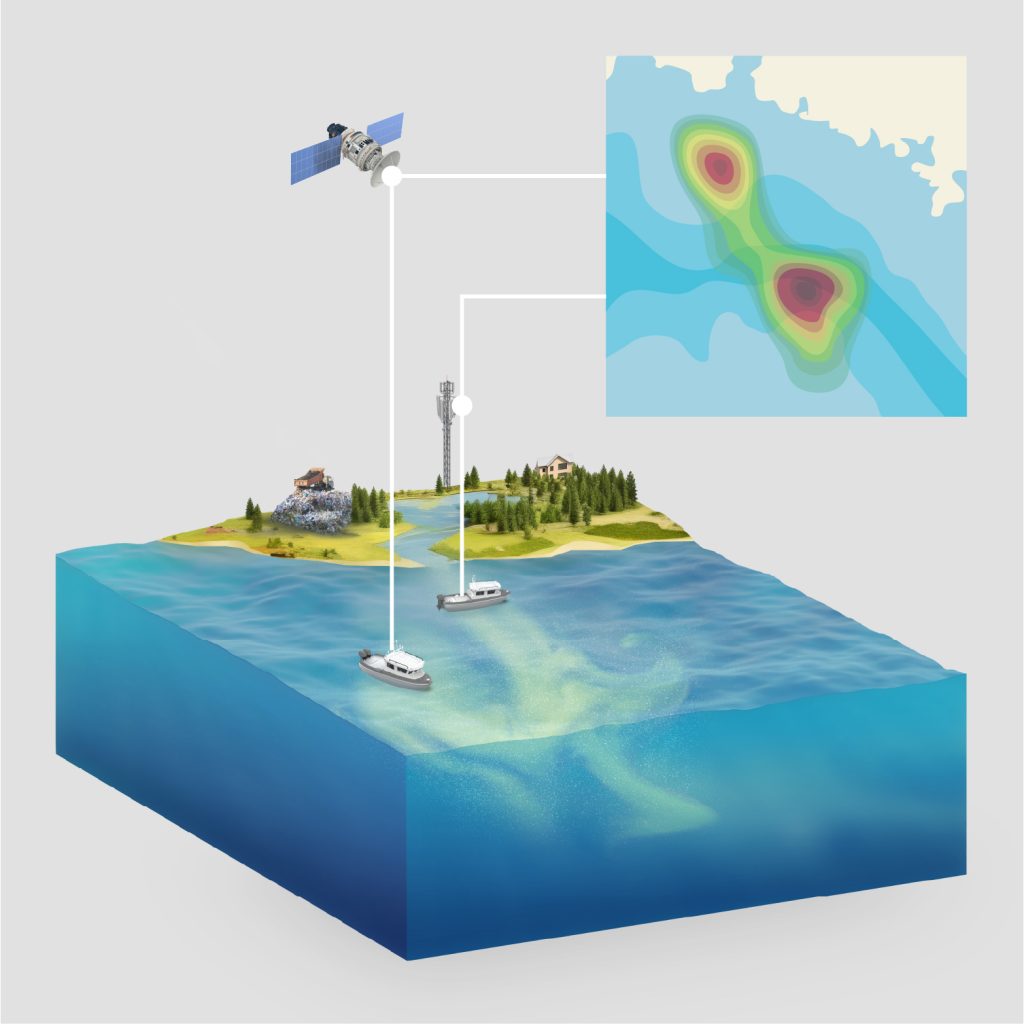
Triple Ring supported the development of a portable microplastics monitoring system designed to measure plastic particle concentrations in aqueous environmental samples. The system was developed to enable field-based quantification of microplastics and support environmental research initiatives.
The resulting platform integrates particle detection and separation technologies into a miniaturized unit capable of replacing larger laboratory-based instrumentation while maintaining measurement accuracy under real-world conditions.
Challenge
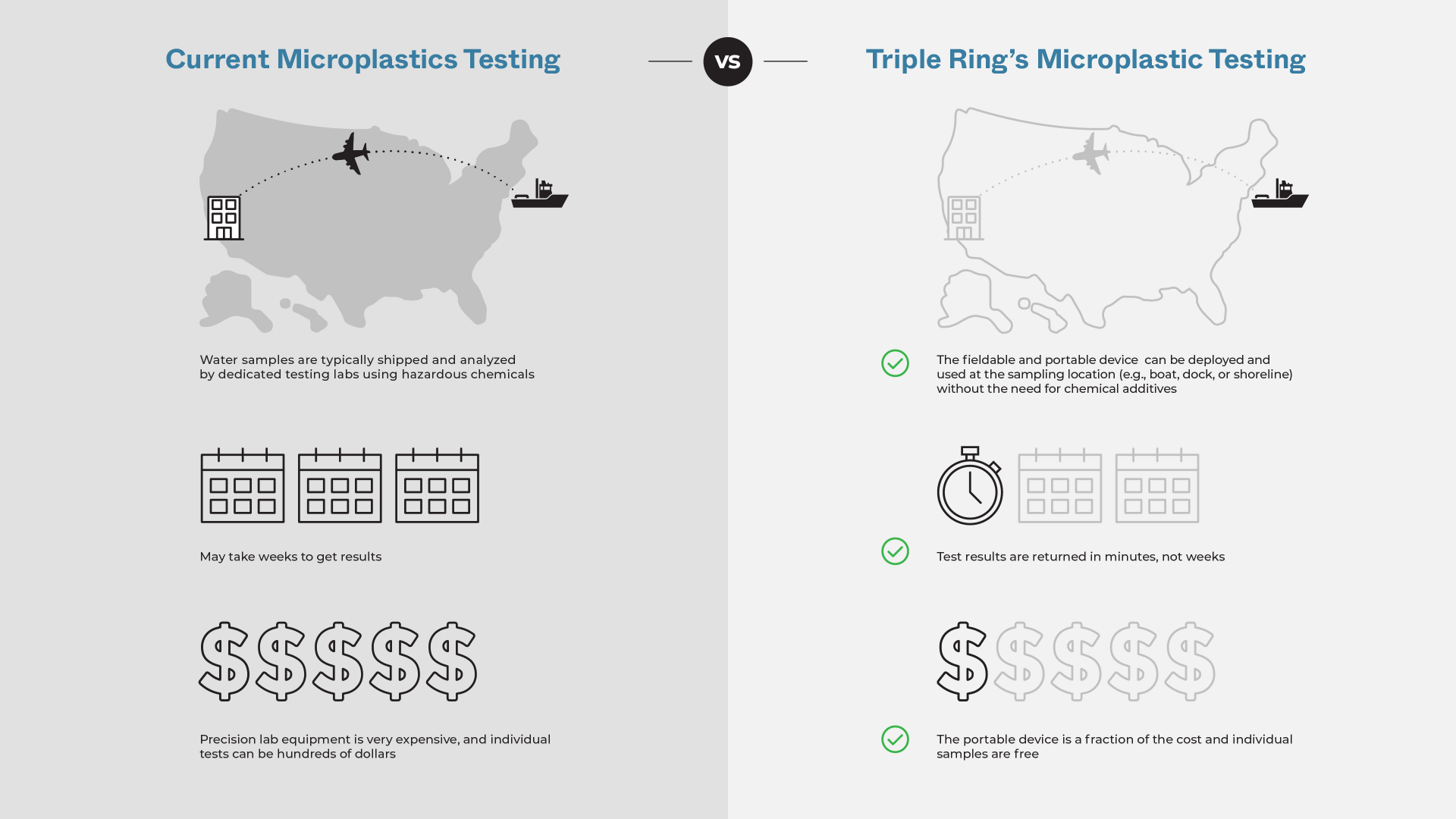
Environmental researchers required a field-deployable system capable of accurately detecting and quantifying microplastics in complex environmental samples. Traditional benchtop instruments were not suitable for field use due to size, cost, and sensitivity to environmental conditions.
The system needed to function reliably in the presence of common interferents such as air bubbles, biological materials, sand, and other particulate matter. Achieving consistent performance under these variable conditions required robust system integration and miniaturization.
Ocean Microplastics Monitor
Overview
Triple Ring supported the development of a portable microplastics monitoring system designed to measure plastic particle concentrations in aqueous environmental samples. The system was developed to enable field-based quantification of microplastics and support environmental research initiatives.
The resulting platform integrates particle detection and separation technologies into a miniaturized unit capable of replacing larger laboratory-based instrumentation while maintaining measurement accuracy under real-world conditions.
Challenge
Environmental researchers required a field-deployable system capable of accurately detecting and quantifying microplastics in complex environmental samples. Traditional benchtop instruments were not suitable for field use due to size, cost, and sensitivity to environmental conditions.
The system needed to function reliably in the presence of common interferents such as air bubbles, biological materials, sand, and other particulate matter. Achieving consistent performance under these variable conditions required robust system integration and miniaturization.
Client
Various
Practice Areas
Core Disciplines
Solution
Triple Ring collaborated with research partners to integrate sensing technologies into a compact and ruggedized platform capable of supporting field-based environmental monitoring. Engineering efforts focused on miniaturizing complex instrumentation while maintaining detection accuracy across diverse environmental samples.
Technical work included:
- Designing miniaturized detection systems that replaced benchtop instrumentation
- Developing algorithms to distinguish plastics from environmental interferents
- Integrating sampling and analysis components into a portable architecture
- Validating system performance under field-relevant conditions

Outcome
Triple Ring delivered a portable microplastics monitoring system capable of operating outside traditional laboratory environments. The system successfully demonstrated reliable particle detection and measurement in real-world environmental conditions.
The field-ready platform enabled successful demonstrations to research partners and funding organizations, supporting ongoing efforts to advance environmental monitoring technologies and improve understanding of microplastic pollution.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined miniaturized sensing system design, particle detection and separation technologies, and ruggedized hardware engineering to develop a portable microplastics monitor capable of replacing laboratory instrumentation in real-world field environments.
Erick and Sheila collaborated with many talented colleagues across Triple Ring on this project.

Erick Blankenberg
Electro-Mechanical Systems Engineering
Erick Blankenberg contributes to the development of electro-mechanical systems with a focus on controls and optimization. His work supports software and hardware integration for imaging and instrumentation technologies, helping teams refine system performance through coordinated design and testing.

Sheila Hemami, PhD
Electrical Engineering & Enviromental Sciences
Dr. Sheila Hemami brings interdisciplinary teams together to develop technologies that address complex environmental and healthcare challenges. Her work focuses on early-stage innovation and cross-sector collaboration, helping transform emerging ideas into solutions that are practical, deployable, and sustainable.


Robotic Radiation Therapy
Client
Empyrean Medical Systems
Practice Areas
Core Disciplines
Overview
Empyrean Medical Systems worked with Triple Ring to design and develop a compact, robotically guided intra-operative radiation therapy device. The system was engineered to deliver targeted radiation therapy within surgical environments while maintaining a strong focus on usability and patient-centered design.
The resulting platform combined robotic guidance, precision radiation delivery, and mobile system architecture to support flexible clinical workflows and improve intra-operative treatment capabilities.
Challenge
Empyrean required development of a compact, mobile radiation therapy system capable of delivering low-energy radiation with precise beam directionality during surgical procedures. The system needed to maintain high performance standards while remaining easy to operate within the constraints of clinical environments.
In addition to performance requirements, the platform required integration of multiple complex subsystems, including custom x-ray sources, beam steering electronics, and robotic positioning components. The development effort also required preparation of a complete design package supporting regulatory submission.
Robotic Radiation Therapy
Overview
Empyrean Medical Systems worked with Triple Ring to design and develop a compact, robotically guided intra-operative radiation therapy device. The system was engineered to deliver targeted radiation therapy within surgical environments while maintaining a strong focus on usability and patient-centered design.
The resulting platform combined robotic guidance, precision radiation delivery, and mobile system architecture to support flexible clinical workflows and improve intra-operative treatment capabilities.
Challenge
Empyrean required development of a compact, mobile radiation therapy system capable of delivering low-energy radiation with precise beam directionality during surgical procedures. The system needed to maintain high performance standards while remaining easy to operate within the constraints of clinical environments.
In addition to performance requirements, the platform required integration of multiple complex subsystems, including custom x-ray sources, beam steering electronics, and robotic positioning components. The development effort also required preparation of a complete design package supporting regulatory submission.
Client
Empyrean Medical Systems
Practice Areas
Core Disciplines


Solution
Triple Ring collaborated with Empyrean throughout the full product development lifecycle, from concept generation through system integration and clinical validation. The engineering effort focused on delivering precise radiation delivery capabilities while maintaining usability and manufacturability.
Development priorities included:
- Designing custom x-ray source technologies supporting 3D beam directionality
- Applying Monte Carlo simulations to validate radiation delivery performance
- Integrating robotic motion systems supporting accurate positioning
- Preparing documentation supporting FDA regulatory submission
Outcome
Triple Ring delivered a fully integrated radiation therapy system that was verified, clinically validated, and submitted to the U.S. Food and Drug Administration (FDA) for 510(k) clearance. The resulting platform supported regulatory approval and demonstrated reliable clinical performance.
Following regulatory submission, the system design was successfully transferred to manufacturing and launched into the market. The completed platform enabled advancement of intra-operative radiation therapy capabilities and supported commercialization of the robotic radiation delivery system.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined radiation physics, custom x-ray source development, robotic systems integration, and regulatory engineering expertise to deliver a fully verified, FDA-submitted intra-operative radiation therapy platform from concept through market launch.
Chris and Barry collaborated with many talented colleagues across Triple Ring and Empyrean Medical Systems on this project.

Chris Mitchell, PhD
Bio and Electrical Engineering & Program Management
Dr. Chris Mitchell brings deep experience leading multidisciplinary teams developing complex imaging and medical device systems. His work focuses on guiding technical programs from concept through implementation, helping translate advanced technologies into reliable, real-world solutions.

Barry Wood
Biomedical & Mechanical Engineering
Barry Wood develops mechanical and biomedical systems with a focus on design and performance analysis. His work supports the creation of robust solutions for complex applications, contributing to dependable system function from concept through implementation.

User-Centric Breast Imaging
Client
Malcova
Practice Areas
Core Disciplines
Overview
Malcova engaged Triple Ring to redesign and advance a novel breast imaging system capable of delivering full 3D imaging at ultra-low radiation dose while improving patient comfort. The system was designed to expand anatomical coverage beyond what is achievable with standard breast CT technologies.
The resulting platform introduced a gantry-free imaging architecture that enabled flexible source and detector motion, supporting advanced imaging capabilities and improved patient-centered workflows.
Challenge
Malcova developed an early prototype demonstrating the potential for improved breast imaging performance and patient experience. However, the prototype required a comprehensive redesign to support full functionality, human testing, and eventual regulatory submission.
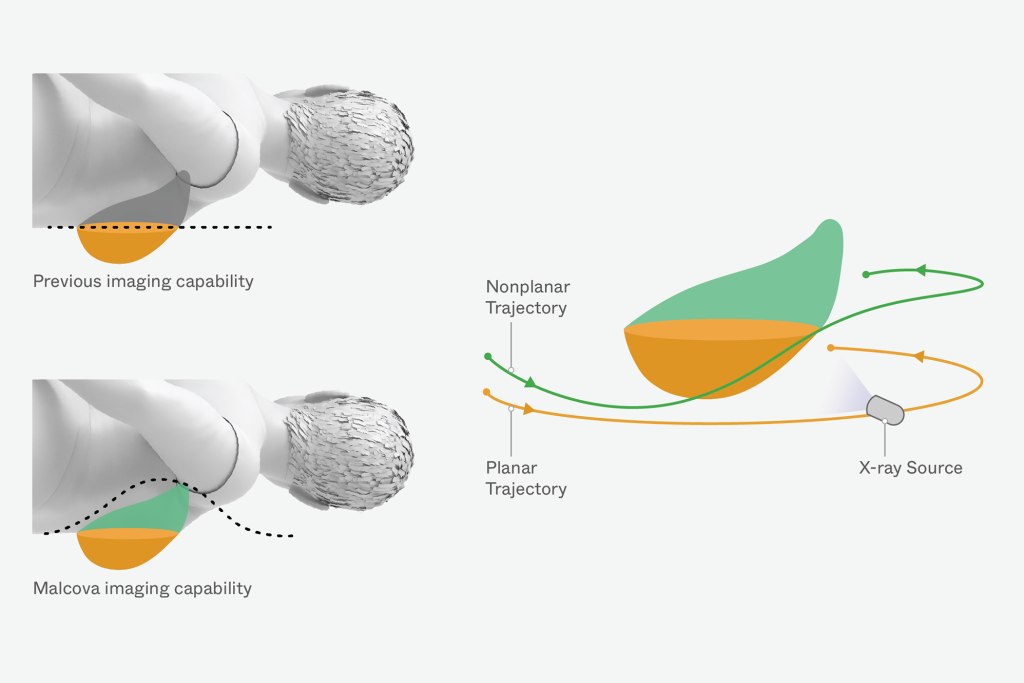
Unlike conventional CT systems that rely on fixed gantry configurations, Malcova’s system required independent movement of the x-ray source and detector to achieve full 3D imaging. This architecture introduced complex synchronization challenges that directly impacted image quality and system reliability. Addressing these challenges required advanced modeling, precise engineering, and deep domain expertise in robotics and x-ray imaging.
User-Centric Breast Imaging
Overview
Malcova engaged Triple Ring to redesign and advance a novel breast imaging system capable of delivering full 3D imaging at ultra-low radiation dose while improving patient comfort. The system was designed to expand anatomical coverage beyond what is achievable with standard breast CT technologies.
The resulting platform introduced a gantry-free imaging architecture that enabled flexible source and detector motion, supporting advanced imaging capabilities and improved patient-centered workflows.
Challenge
Malcova developed an early prototype demonstrating the potential for improved breast imaging performance and patient experience. However, the prototype required a comprehensive redesign to support full functionality, human testing, and eventual regulatory submission.
Unlike conventional CT systems that rely on fixed gantry configurations, Malcova’s system required independent movement of the x-ray source and detector to achieve full 3D imaging. This architecture introduced complex synchronization challenges that directly impacted image quality and system reliability. Addressing these challenges required advanced modeling, precise engineering, and deep domain expertise in robotics and x-ray imaging.
Client
Malcova
Practice Areas
Core Disciplines
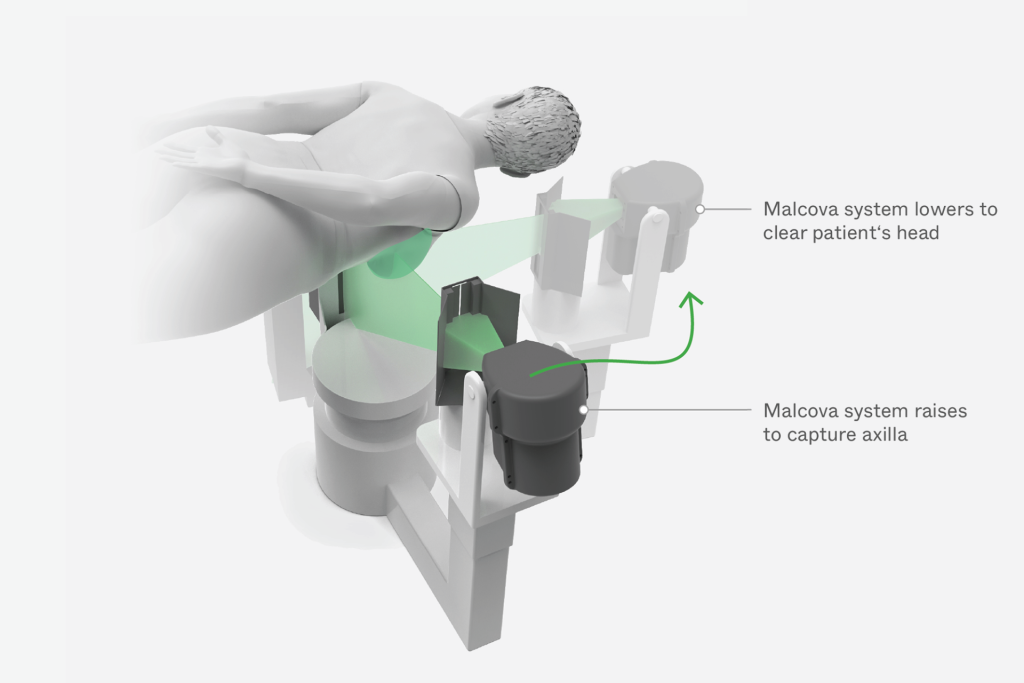
Solution
Triple Ring applied expertise in robotics, x-ray imaging, and simulation-driven design to redesign the imaging platform and enable flexible system operation. The development effort focused on resolving synchronization challenges while enabling exploration of the system’s full imaging capability.
Development focused on:
- Designing coordinated motion control systems supporting gantry-free imaging
- Applying simulation tools to resolve synchronization and trajectory challenges
- Integrating robotics technologies enabling precise source-detector alignment
- Developing control strategies that preserved image fidelity during dynamic motion
- Optimizing imaging workflows to improve patient comfort and accessibility

Outcome
Triple Ring delivered a redesigned CT imaging platform capable of supporting flexible motion and advanced imaging workflows. The resulting system enabled comprehensive exploration of the technology’s design space while maintaining reliable imaging performance.
The completed platform positioned Malcova to advance its innovative breast imaging technology toward clinical validation and future regulatory development, supporting improved imaging coverage and enhanced patient experience.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined robotics, x-ray imaging physics, advanced simulation, and motion control expertise to redesign a gantry-free breast imaging platform — resolving complex synchronization challenges and enabling full 3D imaging at ultra-low radiation dose with expanded anatomical coverage.
Tobias collaborated with many talented colleagues across Triple Ring on this project.

Tobias Funk, PhD
Experimental Physics & Instrumentation
Dr. Tobias Funk develops advanced instrumentation that applies ionizing radiation to scientific and medical challenges. His work spans imaging, simulation, and system design, helping translate complex physical principles into practical technologies used in real-world environments.

At-Home Quantitative Diagnostics
Client
BARDA
Practice Areas
Core Disciplines
Overview
The Biomedical Advanced Research and Development Authority (BARDA) selected Triple Ring to design, build, and test a low-cost quantitative biomarker detection platform intended for at-home and low-resource healthcare settings. The system was developed to support multiplexed biomarker analysis using compact, user-friendly instrumentation.
The resulting platform integrates biological sensing, embedded electronics, and optical detection technologies into a portable diagnostic system designed to support lab-at-home, point-of-care, and direct-to-consumer workflows.
Challenge
BARDA identified the need for a low-cost diagnostic platform capable of delivering quantitative biomarker measurements outside traditional laboratory environments. The system needed to support multiplexed testing while remaining accessible for use in resource-limited settings and CLIA-waived environments.
Achieving this capability required integration of complex biological, optical, and electronic subsystems into a compact and manufacturable design. The platform also needed to extend the measurable range of lateral flow immunoassays while maintaining usability and cost targets suitable for broad deployment.
At-Home Quantitative Diagnostics
Overview
The Biomedical Advanced Research and Development Authority (BARDA) selected Triple Ring to design, build, and test a low-cost quantitative biomarker detection platform intended for at-home and low-resource healthcare settings. The system was developed to support multiplexed biomarker analysis using compact, user-friendly instrumentation.
The resulting platform integrates biological sensing, embedded electronics, and optical detection technologies into a portable diagnostic system designed to support lab-at-home, point-of-care, and direct-to-consumer workflows.
Challenge
BARDA identified the need for a low-cost diagnostic platform capable of delivering quantitative biomarker measurements outside traditional laboratory environments. The system needed to support multiplexed testing while remaining accessible for use in resource-limited settings and CLIA-waived environments.
Achieving this capability required integration of complex biological, optical, and electronic subsystems into a compact and manufacturable design. The platform also needed to extend the measurable range of lateral flow immunoassays while maintaining usability and cost targets suitable for broad deployment.
Client
BARDA
Practice Areas
Core Disciplines


Solution
Triple Ring applied multidisciplinary expertise across biological sciences, engineering, and embedded systems to develop a scalable diagnostic platform capable of delivering quantitative results from multiplexed lateral flow assays.
Development efforts focused on:
- Designing multiplexed lateral flow assay (LFA) detection systems capable of quantifying biomarker concentrations
- Integrating biological, electrical, mechanical, optical, and embedded software subsystems into a unified platform
- Developing workflows suitable for CLIA-waived, point-of-care, and at-home diagnostic environments
- Extending the quantitative measurement range beyond that of conventional lateral flow assay readers
- Conducting system-level integration and verification to support development of a handheld diagnostic platform
Outcome
Triple Ring delivered a functional multiplexed diagnostic platform capable of quantifying biomarker concentrations across multiple test channels. The system demonstrated improved quantitative measurement range compared to conventional lateral flow assay readers.
The platform supported deployment across diverse clinical environments, including at-home and point-of-care settings. Ongoing development efforts include system integration and testing of a handheld version designed to further expand accessibility and usability in remote and low-resource healthcare environments.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined biological sciences, optical detection, embedded electronics, and diagnostic platform engineering to develop a low-cost multiplexed biomarker detection system capable of delivering quantitative lab-grade results in at-home and resource-limited healthcare settings.
Rachel and Chris collaborated with many talented colleagues across Triple Ring and BARDA on this project.

Rachel Gerver, PhD
Bioengineering & Applied Science and Systems
Dr. Rachel Gerver advances microfluidic and point-of-care technologies from early development into real-world application. Her work spans technical leadership and systems development, helping teams bring complex innovations to market where they can deliver meaningful impact.

Chris Todd
Mechanical Engineering
Chris Todd focuses on biomedical diagnostics and precision instrumentation, integrating fluidics, optics, and mechanical systems. His work supports the development of regulated medical technologies from early concepts through manufacturing, helping teams deliver reliable diagnostic solutions at scale.
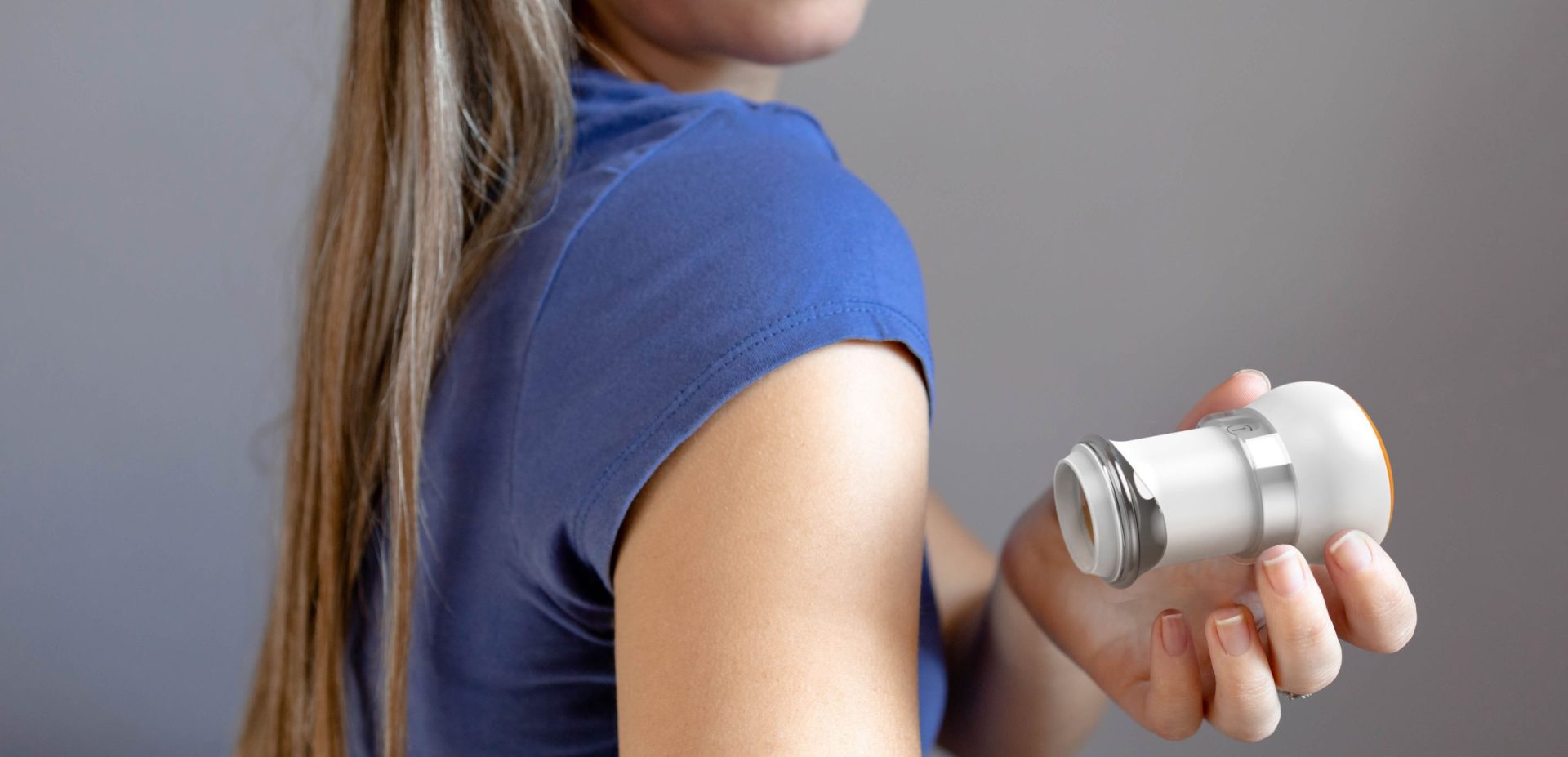
Microneedle Patch Applicator
Client
Confidential
Practice Areas
Core Disciplines
Overview
The client selected Triple Ring to design a reusable applicator supporting the delivery of a novel micro-needle patch for transdermal drug administration. The system was intended for patient-administered use in home settings and required consistent mechanical performance to ensure reliable drug delivery.
The resulting combination product integrated a reusable applicator with a micro-needle patch system designed to enable uniform pressure application, supporting consistent adhesion and controlled dosing across diverse patient populations.
Challenge
The client developed a microneedle-based transdermal patch that required significantly greater uniformity of application pressure compared to traditional adhesive patches. Achieving reliable drug delivery required development of a reusable applicator capable of delivering consistent mechanical force during use.
The applicator needed to function effectively across a wide range of skin types, including variations in thickness, age, moisture content, and anatomical placement. In addition to performance requirements, the design needed to meet durability expectations for repeated home use while maintaining low production cost.
Microneedle Patch Applicator
Overview
The client selected Triple Ring to design a reusable applicator supporting the delivery of a novel micro-needle patch for transdermal drug administration. The system was intended for patient-administered use in home settings and required consistent mechanical performance to ensure reliable drug delivery.
The resulting combination product integrated a reusable applicator with a micro-needle patch system designed to enable uniform pressure application, supporting consistent adhesion and controlled dosing across diverse patient populations.
Challenge
The client developed a microneedle-based transdermal patch that required significantly greater uniformity of application pressure compared to traditional adhesive patches. Achieving reliable drug delivery required development of a reusable applicator capable of delivering consistent mechanical force during use.
The applicator needed to function effectively across a wide range of skin types, including variations in thickness, age, moisture content, and anatomical placement. In addition to performance requirements, the design needed to meet durability expectations for repeated home use while maintaining low production cost.
Client
Confidential
Practice Areas
Core Disciplines

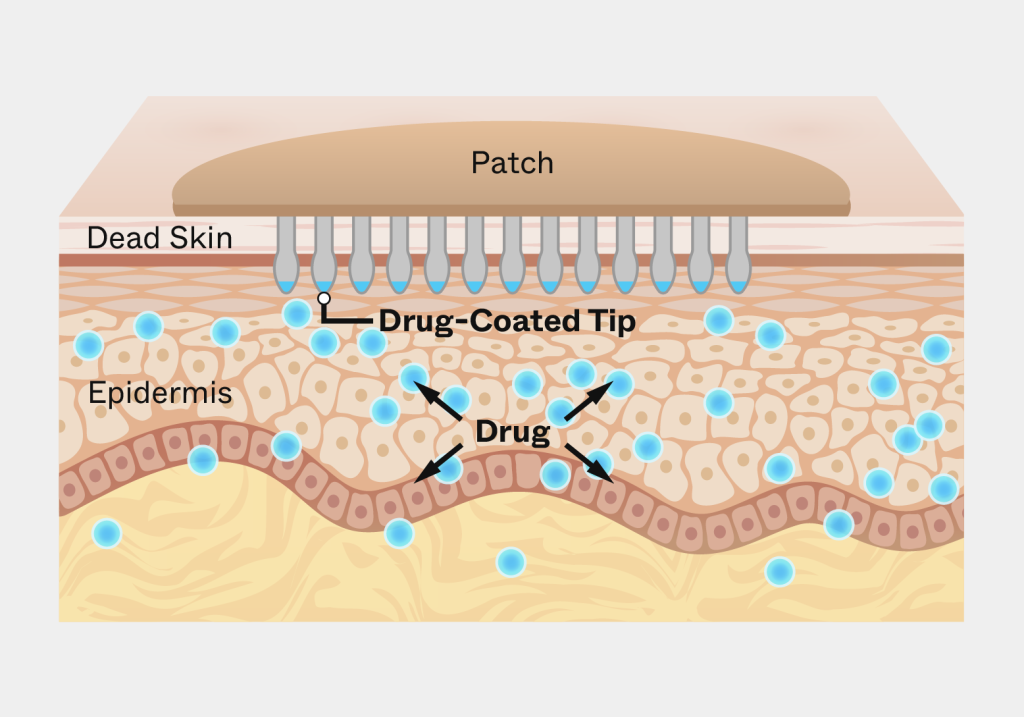
Solution
Triple Ring applied interdisciplinary expertise across materials science, mechanical engineering, and life sciences to design an applicator capable of delivering controlled pressure during patch placement. The development effort focused on optimizing performance across diverse user conditions while maintaining manufacturability and reliability.
Key development efforts included:
- Designing precision mechanical systems capable of delivering uniform pressure during patch application
- Selecting materials optimized for durability, manufacturability, and patient-safe use
- Engineering applicator geometry to accommodate variation in skin thickness, moisture, and elasticity
- Validating device performance across simulated patient-use conditions
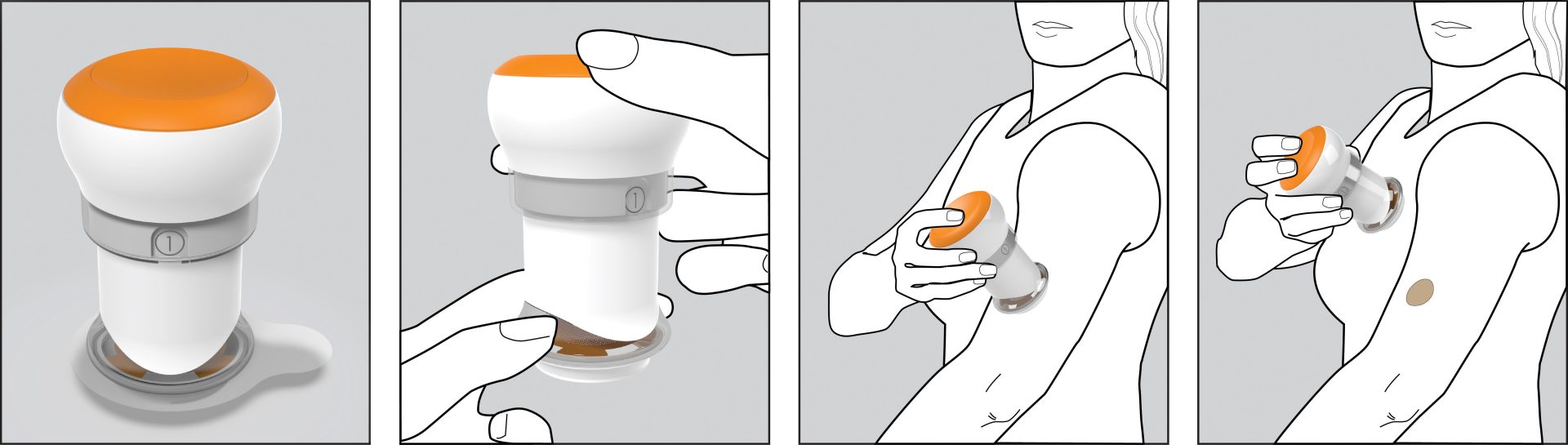
Outcome
Triple Ring delivered a reusable applicator integrated with a micro-needle array patch, forming a single-entity combination product designed for intracutaneous drug delivery. The system enabled rapid drug absorption while supporting consistent dosing performance.
The completed applicator enabled the client to advance into clinical trials and demonstrate the effectiveness of the technology to healthcare stakeholders. The underlying technology was ultimately acquired by a vaccine manufacturer, supporting continued development and commercialization.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined mechanical engineering, materials science, and life sciences expertise to design a reusable applicator capable of delivering consistent, uniform pressure across diverse patient skin types — enabling reliable transdermal drug delivery in home settings.
Walt and Gabe collaborated with many talented colleagues across Triple Ring on this project.

Walt Cecka
Medical Device Design & Translational Medicine
Walt Cecka brings decades of experience translating novel medical technologies into first-to-market products. He works closely with innovators to shape early concepts into structured development programs that advance patient care across a wide range of clinical applications.

Gabriel Chow, PhD
Materials Characterization & Development
Dr. Gabe Chow specializes in materials characterization and mechanical system development across macro to nanoscale environments. His work supports the design, testing, and reliability of advanced materials and mechanical systems, helping translate innovative concepts into durable, real-world solutions.


Kuri, the Nimble Companion Robot
Client
Mayfield Robotics
Practice Areas
Core Disciplines
Overview
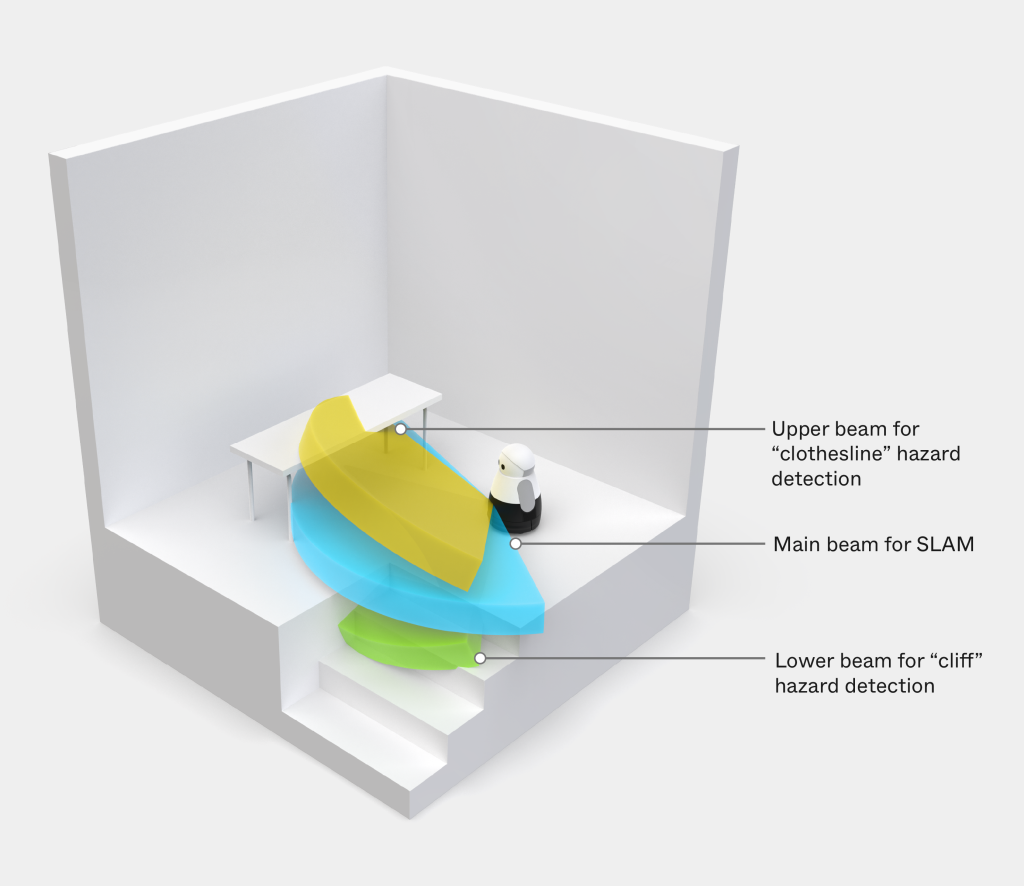
Mayfield Robotics collaborated with Triple Ring to design and develop an optical guidance system for Kuri, a home companion robot designed to safely navigate and interact within household environments. The system required advanced sensing capabilities to support navigation, hazard detection, and environmental awareness.
The resulting optical platform integrated multiple sensing technologies into a compact architecture capable of supporting simultaneous localization and mapping (SLAM) while meeting strict safety and cost requirements for consumer deployment.
Challenge
Kuri required a sophisticated optical system capable of performing multiple functions, including hazard detection, navigation, and real-time environmental mapping. The system needed to maintain high performance in dynamic home environments while remaining safe for use around humans and pets.
Additionally, the integrated optics needed to meet strict eye-safety certification requirements and support reliable operation under consumer use conditions. Designing this system required combining complex optical components within a compact and manufacturable architecture while meeting aggressive launch timelines.
Kuri, the Nimble Companion Robot
Overview
Mayfield Robotics collaborated with Triple Ring to design and develop an optical guidance system for Kuri, a home companion robot designed to safely navigate and interact within household environments. The system required advanced sensing capabilities to support navigation, hazard detection, and environmental awareness.
The resulting optical platform integrated multiple sensing technologies into a compact architecture capable of supporting simultaneous localization and mapping (SLAM) while meeting strict safety and cost requirements for consumer deployment.
Challenge
Kuri required a sophisticated optical system capable of performing multiple functions, including hazard detection, navigation, and real-time environmental mapping. The system needed to maintain high performance in dynamic home environments while remaining safe for use around humans and pets.
Additionally, the integrated optics needed to meet strict eye-safety certification requirements and support reliable operation under consumer use conditions. Designing this system required combining complex optical components within a compact and manufacturable architecture while meeting aggressive launch timelines.
Client
Mayfield Robotics
Practice Areas
Core Disciplines

Solution
Triple Ring applied deep expertise in optical system design and robotics integration to develop a navigation system capable of supporting safe and reliable robot operation. The development effort focused on integrating advanced sensing technologies into a scalable and cost-effective architecture.
Engineering efforts included:
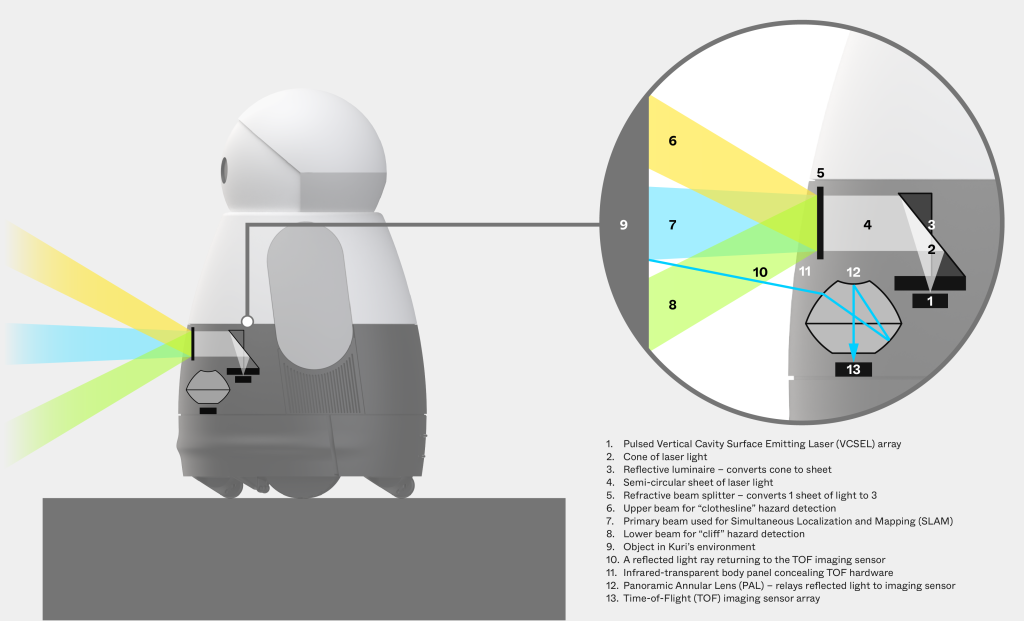
- Designing pulsed vertical cavity surface emitting laser (VCSEL) array systems supporting depth sensing
- Developing panoramic annular lens configurations enabling wide-area environmental awareness
- Integrating time-of-flight imaging sensor arrays for real-time distance measurement
- Engineering optical subsystems compliant with stringent eye-safety requirements
- Supporting system-level integration and validation to meet cost, performance, and production goals

Outcome
Triple Ring delivered an optical navigation system that exceeded cost and performance requirements while achieving certification for eye safety and compliance with UL 3300 standards. The system enabled reliable navigation and safe operation within home environments.
The completed optical system supported the successful commercial launch of Kuri, with hundreds of units delivered to customers. The design generated multiple patent filings and contributed to Kuri being named Best in Show at CES 2017 by leading technology publications.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined optical system design, VCSEL and time-of-flight sensing, robotics integration, and consumer safety certification expertise to develop a compact, eye-safe optical navigation system capable of supporting simultaneous localization, mapping, and hazard detection in dynamic home environments.
Todd collaborated with many talented colleagues across Triple Ring and Mayfield Robotics on this project.

Todd Harris, PhD
Physics & Optical Science
Dr. Todd Harris applies expertise in optics and imaging physics to the development of advanced sensing and illumination technologies. His work combines optical modeling with system design, helping teams translate complex physical principles into dependable, high-performance solutions.

IVD Platform Refresh
Client
Fortune 500 IVD Company
Practice Areas
Core Disciplines
Overview
A Fortune 500 diagnostics manufacturer worked with Triple Ring to modernize a legacy in vitro diagnostic (IVD) platform facing component obsolescence and software limitations. The project focused on refreshing the system architecture while maintaining regulatory equivalency with an existing FDA-cleared product.
The resulting platform replaced obsolete hardware, migrated legacy software, and introduced updated industrial design elements while preserving compatibility with established manufacturing and regulatory pathways.
Challenge
The client faced the obsolescence of critical hardware components, including single-board computers and microcontrollers, within an existing diagnostic platform. Compounding the challenge, original firmware source code was unavailable, and institutional knowledge associated with the system had diminished over time.
In addition to restoring functionality, the refreshed system needed to maintain regulatory equivalence to the original device in order to qualify for a Special 510(k) submission. This requirement demanded careful reverse engineering, system validation, and modernization without introducing unintended performance deviations.
IVD Platform Refresh
Overview
A Fortune 500 diagnostics manufacturer worked with Triple Ring to modernize a legacy in vitro diagnostic (IVD) platform facing component obsolescence and software limitations. The project focused on refreshing the system architecture while maintaining regulatory equivalency with an existing FDA-cleared product.
The resulting platform replaced obsolete hardware, migrated legacy software, and introduced updated industrial design elements while preserving compatibility with established manufacturing and regulatory pathways.
Challenge
The client faced the obsolescence of critical hardware components, including single-board computers and microcontrollers, within an existing diagnostic platform. Compounding the challenge, original firmware source code was unavailable, and institutional knowledge associated with the system had diminished over time.
In addition to restoring functionality, the refreshed system needed to maintain regulatory equivalence to the original device in order to qualify for a Special 510(k) submission. This requirement demanded careful reverse engineering, system validation, and modernization without introducing unintended performance deviations.
Client
Fortune 500 IVD Company
Practice Areas
Core Disciplines


Solution
Triple Ring implemented a structured modernization strategy combining reverse engineering, hardware redesign, and software migration. The development effort focused on preserving functional equivalency while introducing modern components and improving long-term maintainability.
Development efforts included:
- Reverse-engineering legacy hardware and firmware to recover system functionality and requirements
- Migrating existing software to a modern operating system architecture
- Replacing obsolete components while maintaining compatibility with legacy subsystems
- Updating mechanical and electrical subassemblies to support manufacturing continuity
- Preparing validation and documentation packages supporting regulatory equivalence requirements
Outcome
Triple Ring delivered a fully refreshed IVD platform supported by a comprehensive regulatory submission package demonstrating equivalency to the original FDA-cleared device. The updated system maintained functional continuity while addressing long-term hardware and software sustainability.
The refreshed design integrated seamlessly into existing manufacturing workflows, enabling uninterrupted production and maintaining cost targets. The modernization effort positioned the platform for continued market competitiveness while ensuring regulatory compliance and operational reliability.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined reverse engineering, embedded systems, software migration, regulatory strategy, and industrial design expertise to modernize a legacy IVD platform — restoring long-term sustainability while maintaining full regulatory equivalency with the original FDA-cleared device.
Rob and Anne collaborated with many talented colleagues across Triple Ring on this project.

Robert de Saint Phalle
Interdisciplinary Product Design
Robert de Saint Phalle shapes product design strategies that connect engineering, user experience, and emerging technologies. His work helps teams translate complex ideas into thoughtful, functional products that bring new technologies to life.

Anne Preut
Biomedical & Systems Engineering
Anne Preut leads systems engineering efforts that integrate hardware, firmware, assays, and fluidics across complex diagnostic technologies. Her work aligns cross-functional teams throughout the development lifecycle, helping ensure systems perform reliably from early feasibility through production.