Continuous Glucose Monitoring
Client
Dexcom
Practice Areas
Core Disciplines
Overview
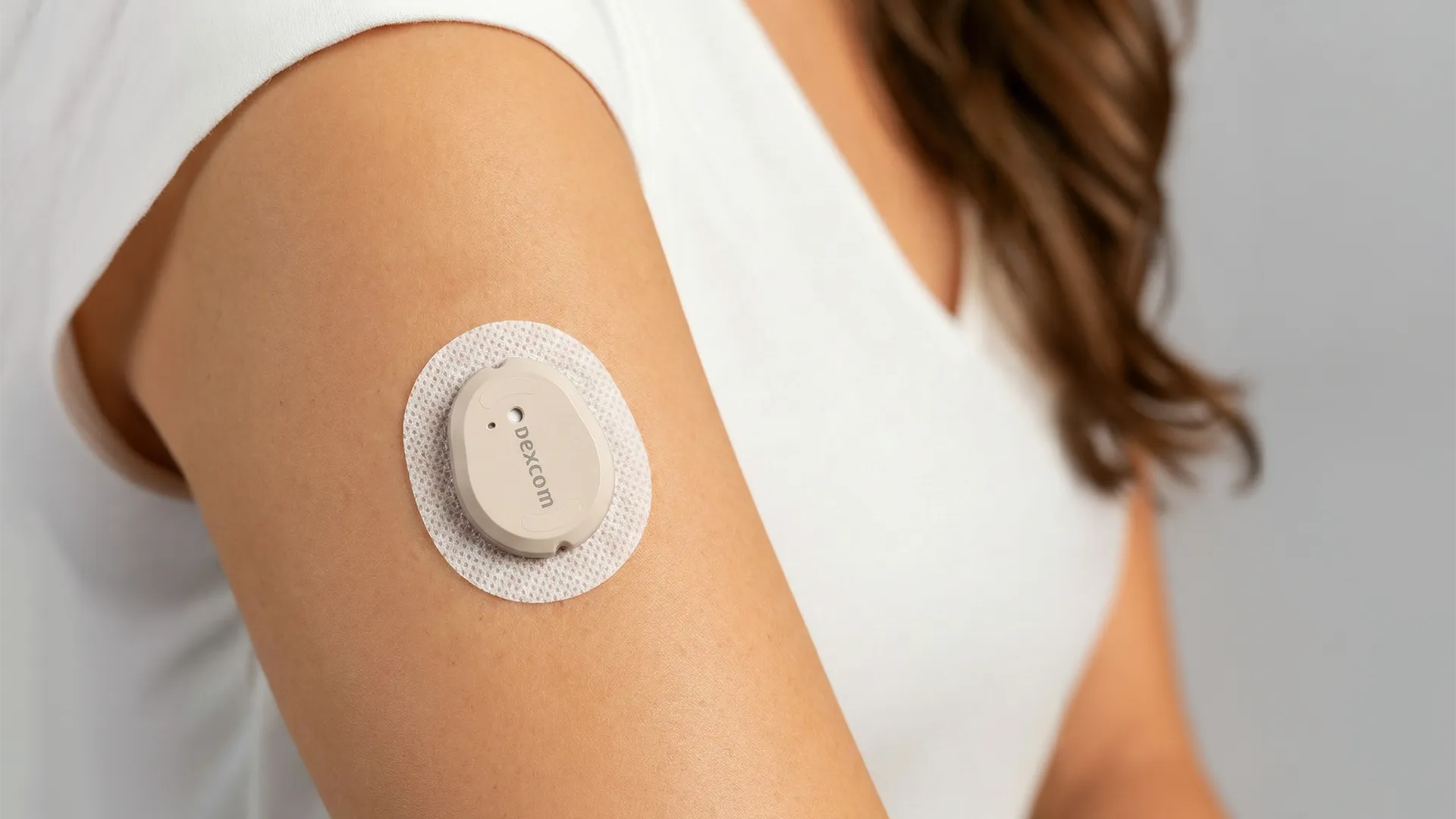
For more than a decade, Triple Ring has partnered with continuous glucose monitoring (CGM) manufacturer Dexcom to support the rapid evolution of product designs in response to customer demands and increasing market competition. Dexcom CGM systems are wearable medical devices that continuously track glucose levels and deliver real-time data to connected devices, enabling improved diabetes management.
Across multiple generations of CGM devices, Triple Ring contributed analytical expertise and design inputs that enabled new features, improved manufacturability, and supported high-volume production. This long-term collaboration helped shorten development timelines and accelerate the release of successive CGM platforms.
Challenge
As Dexcom’s CGM products evolved, market forces required rapid improvements in usability, reliability, and manufacturability while maintaining high performance and production efficiency. The development of next-generation CGM devices required resolving complex design behaviors, managing manufacturing variability, and supporting large-scale production.
Dexcom engaged Triple Ring at critical points in development to address these technical challenges while maintaining aggressive timelines for new product releases.
Continuous Glucose Monitoring
Overview
For more than a decade, Triple Ring has partnered with continuous glucose monitoring (CGM) manufacturer Dexcom to support the rapid evolution of product designs in response to customer demands and increasing market competition. Dexcom CGM systems are wearable medical devices that continuously track glucose levels and deliver real-time data to connected devices, enabling improved diabetes management.
Across multiple generations of CGM devices, Triple Ring contributed analytical expertise and design inputs that enabled new features, improved manufacturability, and supported high-volume production. This long-term collaboration helped shorten development timelines and accelerate the release of successive CGM platforms.
Challenge
As Dexcom’s CGM products evolved, market forces required rapid improvements in usability, reliability, and manufacturability while maintaining high performance and production efficiency. The development of next-generation CGM devices required resolving complex design behaviors, managing manufacturing variability, and supporting large-scale production.
Dexcom engaged Triple Ring at critical points in development to address these technical challenges while maintaining aggressive timelines for new product releases.
Client
Dexcom
Practice Areas
Core Disciplines

Solution
Triple Ring applied deep expertise in device development, mechanism analysis, finite-element simulation, and empirical testing to support the design and optimization of Dexcom’s CGM platforms.
By leveraging system-level modeling and complex empirical characterization, Triple Ring enabled Dexcom to model the effects of manufacturing variability across hundreds of thousands of virtual devices. This approach allowed the identification of unforeseen design sensitivities early in development, when design changes were less costly and more effective.
Development efforts included:
- Designing automated one-touch deployment mechanisms
- Conducting system-level modeling to evaluate design performance
- Developing testing strategies to capture real-world device behavior
- Refining designs to improve manufacturability and yield
Solution
Dexcom, together with Triple Ring, developed and optimized a fully automated one-touch deployment mechanism for the award-winning G6 platform. Five years later, the G7 successor introduced a completely redesigned applicator that combined deployment of the wearable and sensor into a single step, reflecting continued advances in device integration and usability.
Triple Ring’s analytical and empirical strategies resolved undesirable device behaviors, improved model accuracy, and ensured robust product performance. These efforts significantly reduced time to market while enabling reliable, high-volume manufacturing.
The collaboration established Triple Ring as a long-term trusted partner supporting Dexcom’s continued innovation in diabetes management and improving quality of life for millions of patients.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined mechanism analysis, finite-element simulation, system-level modeling, and empirical testing to help Dexcom design and optimize CGM platforms across multiple device generations — shortening timelines and enabling reliable, high-volume manufacturing.
Gabe and Thu collaborated with many talented colleagues across Triple Ring and Dexcom on this project.

Gabriel Chow, PhD
Materials Characterization & Development
Dr. Gabe Chow specializes in materials characterization and mechanical system development across macro to nanoscale environments. His work supports the design, testing, and reliability of advanced materials and mechanical systems, helping translate innovative concepts into durable, real-world solutions.

Thu Nguyen, PhD
Mechanical Systems & Device Development
Dr. Thu Nguyen specializes in human-centered mechanical systems, with expertise in exoskeleton control and movement optimization. Her work focuses on understanding how mechanical design interacts with human performance, helping teams develop solutions that improve function, reliability, and real-world usability.


Real-Time Tissue Oxygenation Status
Client
ViOptix
Practice Areas
Core Disciplines
Overview
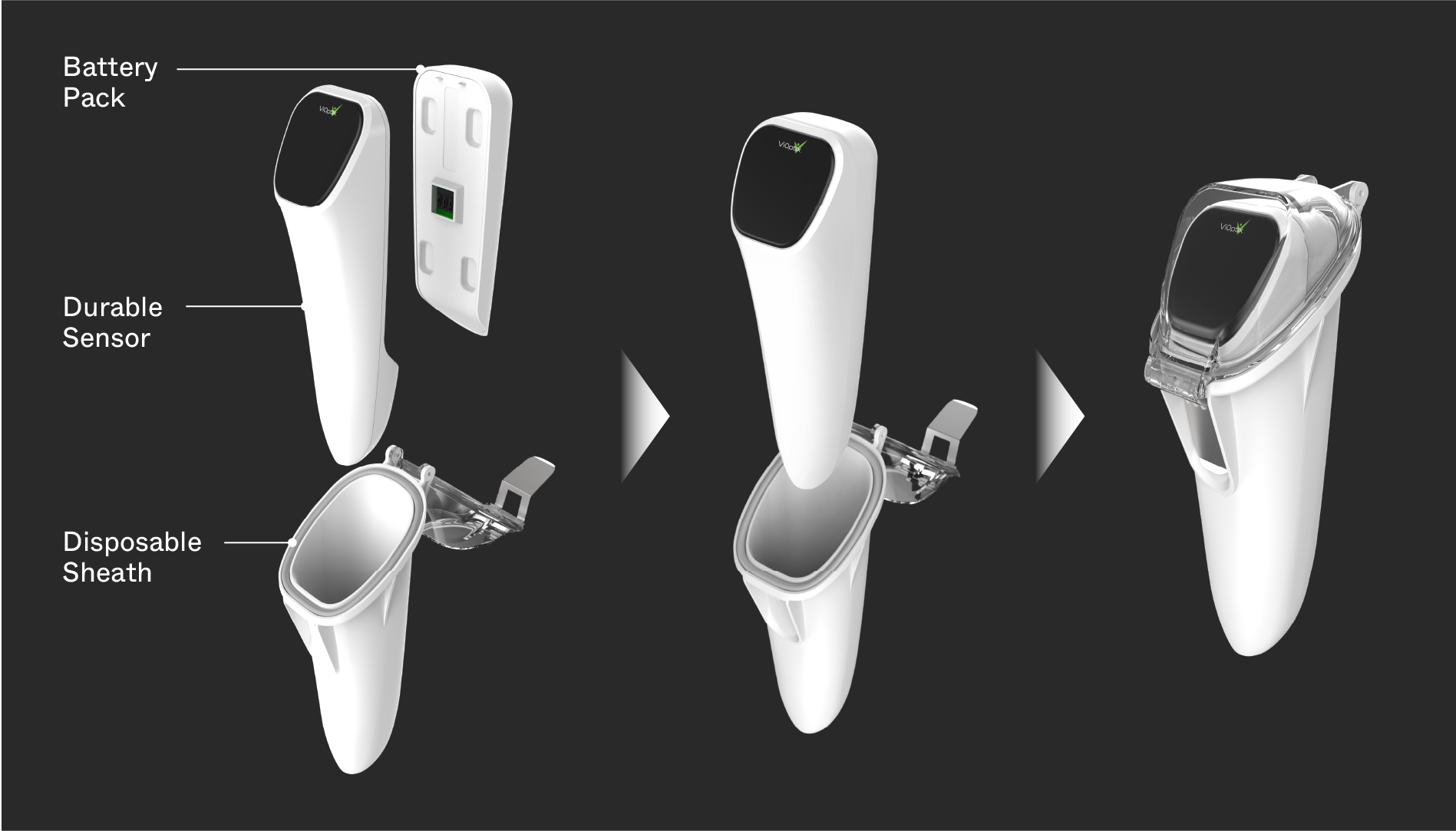
ViOptix collaborated with Triple Ring to design and develop the Intra.Ox™, a non-invasive, handheld tissue oxygenation monitor designed for use inside the surgical theater. The device enables real-time measurement of tissue oxygenation, helping surgeons assess tissue viability and make informed decisions during procedures.
By combining optical sensing technology with advanced algorithms and a portable handheld design, the system supports improved surgical workflows and enhances clinical decision-making during tissue transfer procedures.
Challenge
ViOptix sought to develop a quantitative tissue oxygenation monitor capable of delivering real-time, highly sensitive measurements in a surgical environment. The system needed to be easy to use, reliable, and capable of generating unlimited readings throughout a surgical case.
Key technical challenges included:
- Designing an easy-to-use multi-wavelength tissue oximeter
- Developing smart algorithms to compensate for variable tissue morphologies
- Creating a compact handheld form factor suitable for surgical use
- Supporting reusable packaging and surgical workflows
- Ensuring consistent performance across varying tissue conditions
In addition, the system needed to accurately capture oxygen concentration in resected tissue while minimizing artifacts caused by surrounding tissue components.
Real-Time Tissue Oxygenation Status
Overview
ViOptix collaborated with Triple Ring to design and develop the Intra.Ox™, a non-invasive, handheld tissue oxygenation monitor designed for use inside the surgical theater. The device enables real-time measurement of tissue oxygenation, helping surgeons assess tissue viability and make informed decisions during procedures.
By combining optical sensing technology with advanced algorithms and a portable handheld design, the system supports improved surgical workflows and enhances clinical decision-making during tissue transfer procedures.
Challenge
ViOptix sought to develop a quantitative tissue oxygenation monitor capable of delivering real-time, highly sensitive measurements in a surgical environment. The system needed to be easy to use, reliable, and capable of generating unlimited readings throughout a surgical case.
Key technical challenges included:
- Designing an easy-to-use multi-wavelength tissue oximeter
- Developing smart algorithms to compensate for variable tissue morphologies
- Creating a compact handheld form factor suitable for surgical use
- Supporting reusable packaging and surgical workflows
- Ensuring consistent performance across varying tissue conditions
In addition, the system needed to accurately capture oxygen concentration in resected tissue while minimizing artifacts caused by surrounding tissue components.
Client
ViOptix
Practice Areas
Core Disciplines


Solution
Triple Ring and ViOptix worked side-by-side to invent subsystem- and system-level solutions that enabled reliable real-time tissue oxygenation monitoring. The development effort spanned concept generation through clinical validation, with a strong emphasis on modeling, algorithm design, and usability.
Triple Ring applied deep clinical and technical expertise to accelerate development while supporting the needs of a virtual startup environment. Complex modeling techniques, including Monte Carlo simulations, were used to evaluate optical performance and optimize system behavior early in the design process.
Engineering efforts focused on:
- Designing multi-wavelength optical sensing systems capable of detecting tissue oxygenation levels
- Developing algorithms that compensated for variability in tissue characteristics
- Optimizing handheld system architecture to support surgical usability
- Validating measurement accuracy across simulated clinical environments
Outcome
The ViOptix Intra.Ox technology received FDA clearance after demonstrating its ability to produce accurate, instantaneous estimates of percent saturated oxygen (StO₂) without requiring capital equipment or dye injection.
The device is now used during tissue transfer surgeries to support real-time assessment of tissue viability, enabling earlier intervention and improving both clinical and financial outcomes. The program progressed from concept to clinical validation and FDA 510(k) clearance in approximately 18 months, demonstrating the efficiency of the collaborative development approach.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined advanced optical modeling, Monte Carlo simulation, algorithm development, and user-centered device design to bring a first-of-its-kind handheld tissue oxygenation monitor from concept to FDA clearance in just 18 months.
Jeremy and Nick collaborated with many talented colleagues across Triple Ring and ViOptix on this project.

Nick Hawson
Medical Device Strategy & Program Leadership
With 25 years spanning engineering, program leadership, and innovation consulting, Nick has helped bring groundbreaking medical technologies to market for companies ranging from early-stage startups to global blue-chip firms.

Jeremy Ford, PhD
Biomedical Optics & Physiological Sensing
Dr. Jeremy Ford brings a decade of biomedical optics expertise — from tissue-illumination systems and physiological sensing to optical-thermal simulation and neural inhibition — backed by a Vanderbilt PhD and extensive peer-reviewed research.


Cannabis Breathalyzer System
Client
Hound Labs
Practice Areas
Core Disciplines
Overview
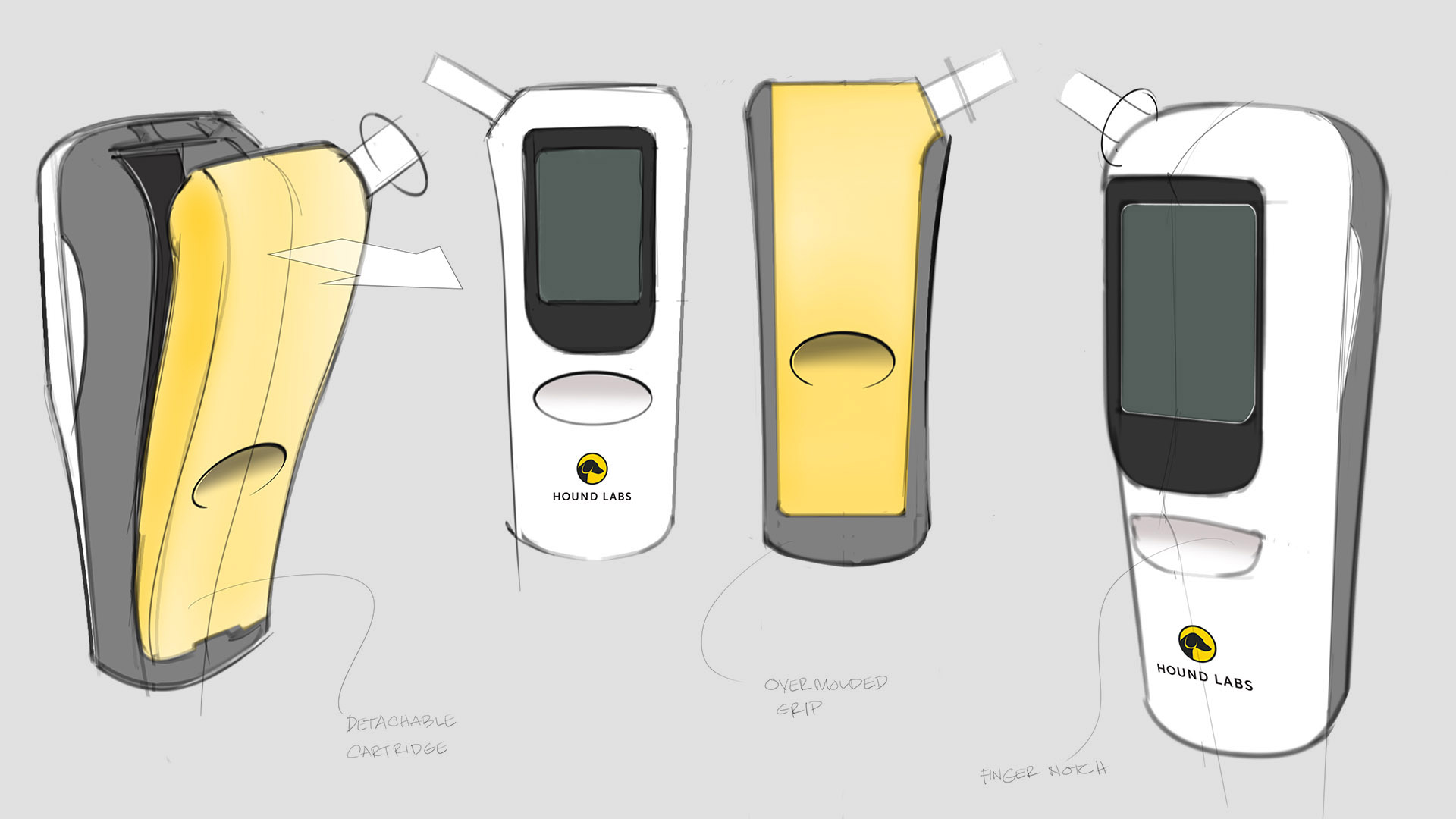
Hound Labs engaged Triple Ring to invent a portable breath analyzer capable of detecting recent marijuana use. The system was developed to support point-of-use testing in law enforcement and workplace environments requiring reliable impairment detection.
Starting from an early concept sketch, Triple Ring collaborated with Hound Labs to develop a high-sensitivity breathalyzer platform combining chemical detection and portable instrumentation technologies.
Challenge
The system needed to detect Δ-9 THC in exhaled breath with clinical-grade sensitivity while maintaining portability and reliability in field conditions.
In addition to engineering challenges, the project required validation of THC pharmacodynamics in breath, including generation of peer-reviewed scientific evidence supporting detection feasibility.
Cannabis Breathalyzer System
Overview
Hound Labs engaged Triple Ring to invent a portable breath analyzer capable of detecting recent marijuana use. The system was developed to support point-of-use testing in law enforcement and workplace environments requiring reliable impairment detection.
Starting from an early concept sketch, Triple Ring collaborated with Hound Labs to develop a high-sensitivity breathalyzer platform combining chemical detection and portable instrumentation technologies.
Challenge
The system needed to detect Δ-9 THC in exhaled breath with clinical-grade sensitivity while maintaining portability and reliability in field conditions.
In addition to engineering challenges, the project required validation of THC pharmacodynamics in breath, including generation of peer-reviewed scientific evidence supporting detection feasibility.
Client
Hound Labs
Practice Areas
Core Disciplines
Solution
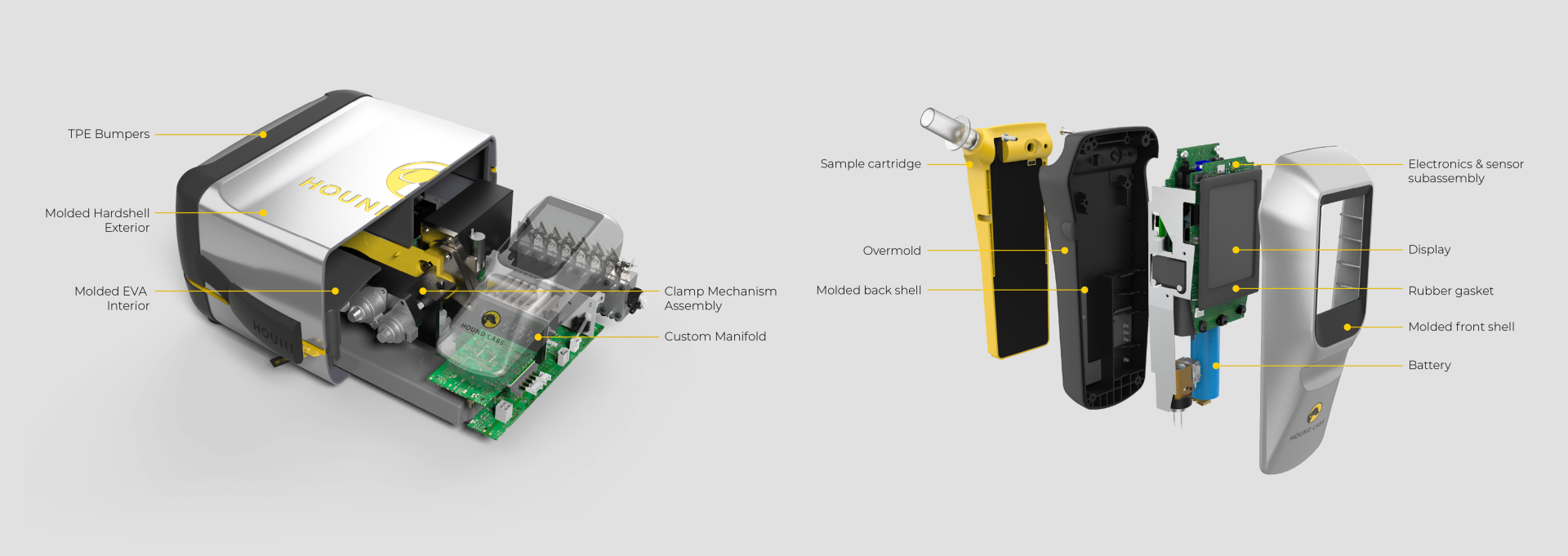
Triple Ring assembled multidisciplinary development teams to design and validate a fully integrated breath analysis platform combining microfluidic sampling and portable device technologies.
Development efforts included:
- Designing a portable breath capture device optimized for reliable sample collection
- Engineering microfluidic lab-on-a-chip cartridges supporting sensitive compound detection
- Integrating control station hardware capable of processing and analyzing captured samples
- Supporting validation activities that produced peer-reviewed pharmacodynamic data
- Developing prototype systems suitable for demonstration, testing, and continued product development

Outcome
Triple Ring delivered fully functioning prototype systems consisting of a breath capture device, control station, and microfluidic cartridges.
The system produced clinical-grade data at the point of use, supporting detection of recent marijuana use and enabling development of a first-in-class breath-based detection platform.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined microfluidic engineering, biological sensing, portable instrumentation, and clinical validation expertise to develop a first-in-class breath-based THC detection platform capable of delivering clinical-grade results at the point of use.
David and Kevin collaborated with many talented colleagues across Triple Ring and Hound Labs on this project.

David Shack, PhD
Mechanical Engineering & Applied Sciences
Dr. David Shack directs the development of complex scientific measurement and analysis systems across multidisciplinary teams. His work integrates algorithms, software, fluidics, and optical technologies, helping advance tools that support precision research and clinical applications.

Kevin Limtao
Biomedical & Systems Engineering
Kevin Limtao connects system requirements, architecture, and integration across complex medical and diagnostic technologies. His work helps teams align design, risk, and performance throughout the development lifecycle, ensuring systems function reliably from early concepts through deployment.

Intra-Operative Tissue Pathology
Client
BlackLight Surgical
Core Disciplines
Overview
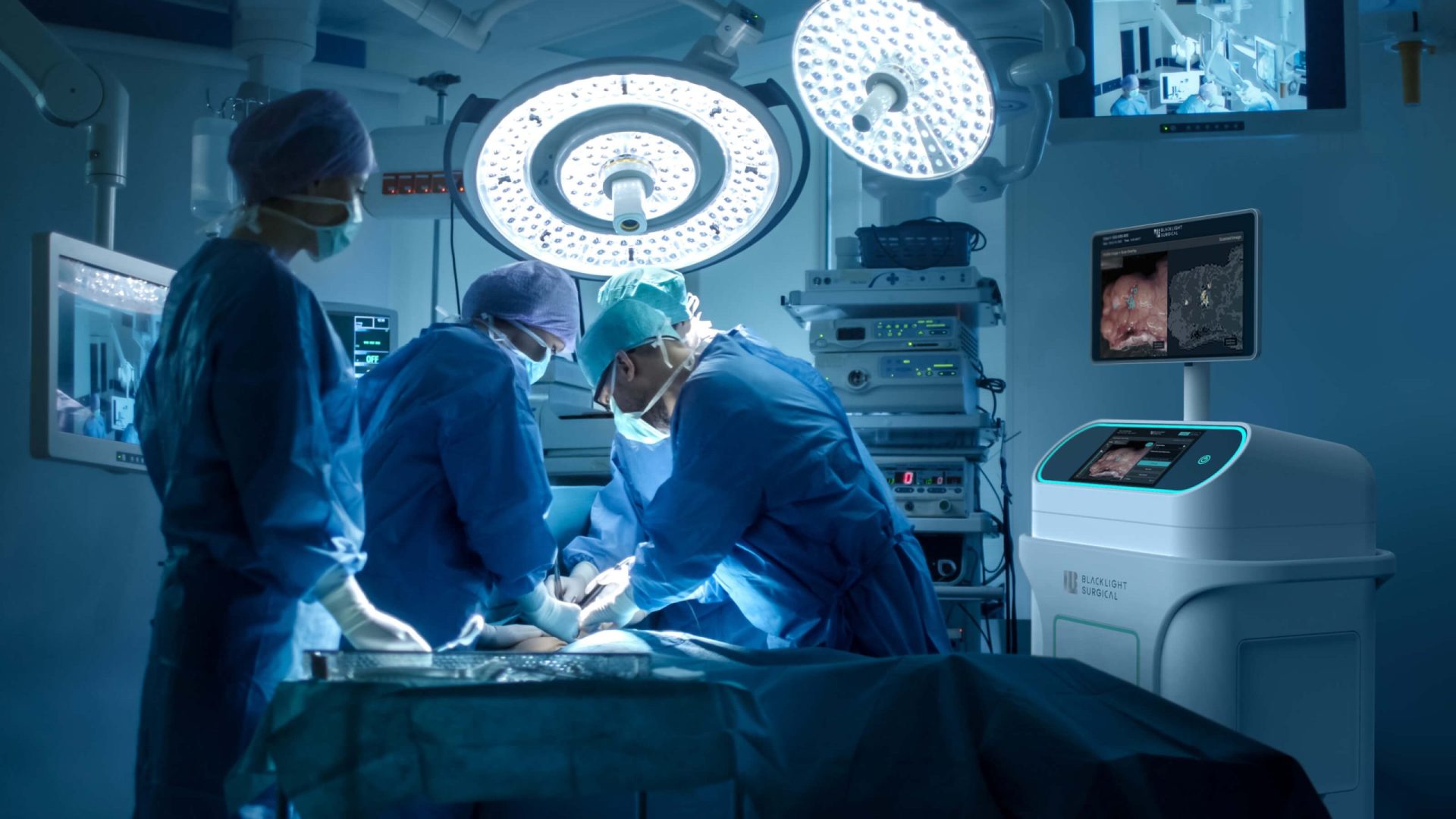
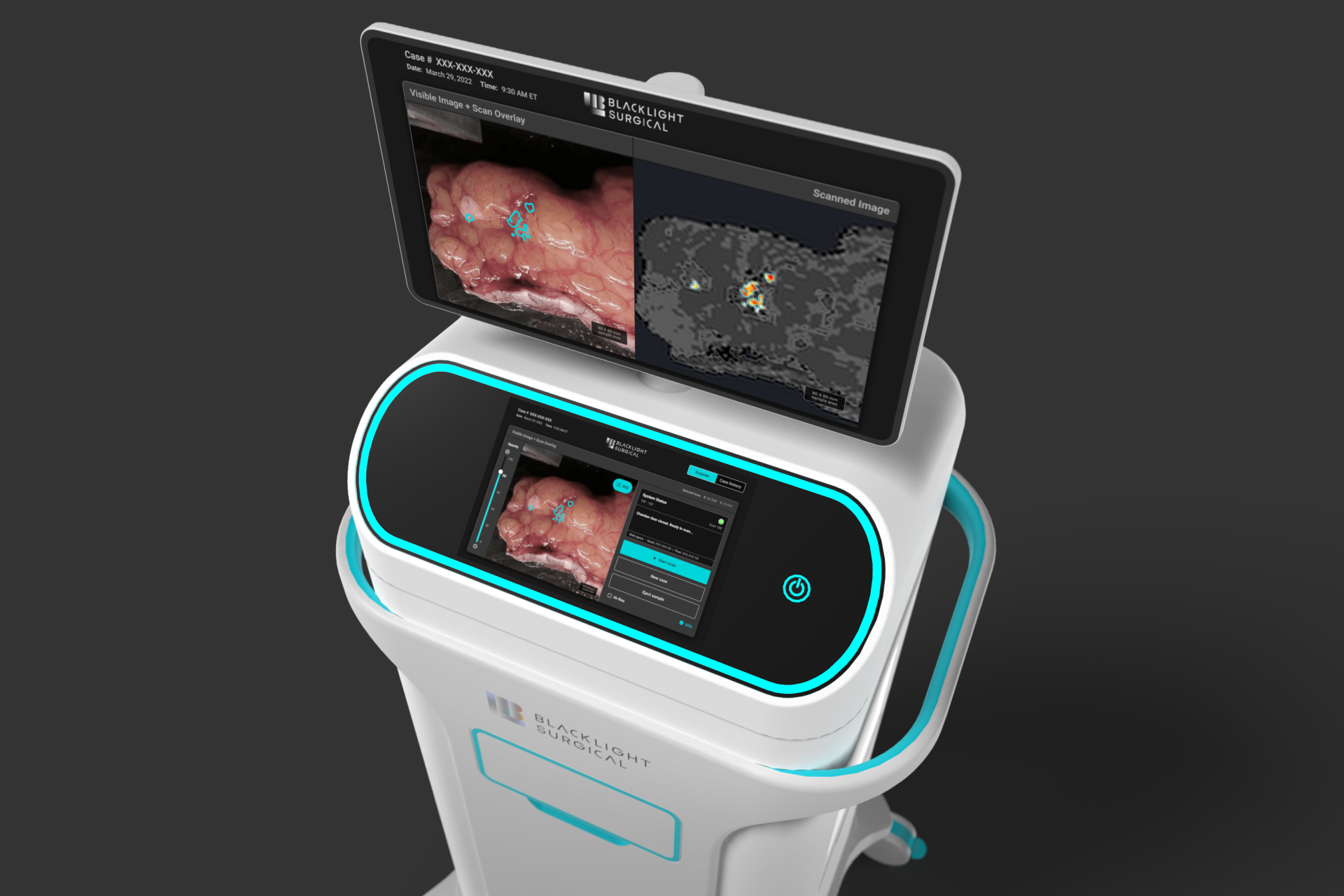
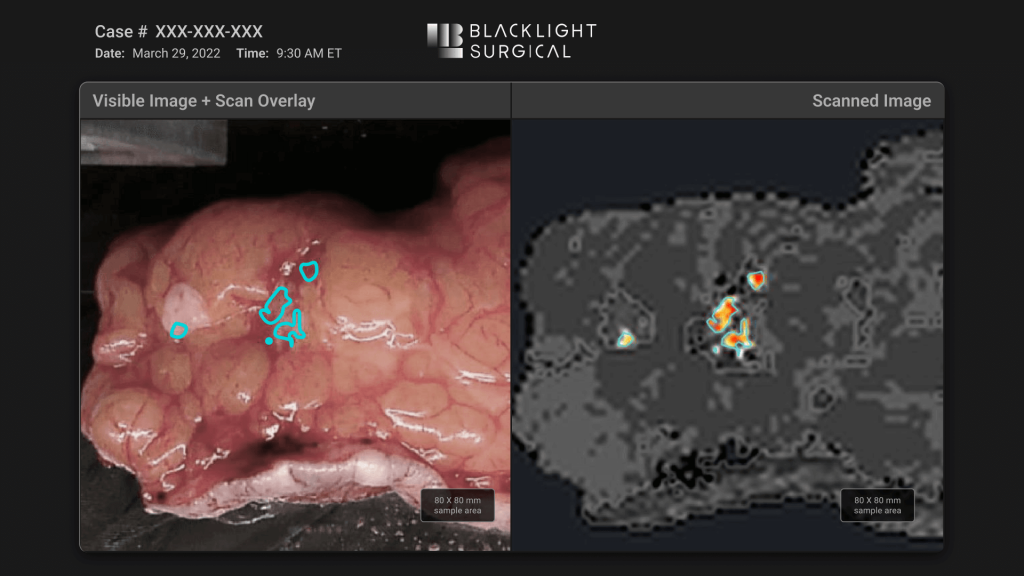
BlackLight Surgical engaged Triple Ring to develop a high-speed biochemical imaging platform designed for intra-operative tissue analysis. The system leveraged picosecond pulsing laser technology and machine learning workflows to enable rapid tissue identification during surgical procedures.
The resulting platform supports real-time clinical decision making by allowing clinicians to distinguish between normal and diseased tissue directly in the operating suite.
Challenge
The system required integration of advanced optical imaging technology capable of performing rapid biochemical analysis during surgery. Reliable performance was required across demanding clinical environments and complex workflows.
Delivering this capability required precise integration of optical, mechanical, software, and machine learning systems into a clinically deployable architecture.
Intra-Operative Tissue Pathology
Overview
BlackLight Surgical engaged Triple Ring to develop a high-speed biochemical imaging platform designed for intra-operative tissue analysis. The system leveraged picosecond pulsing laser technology and machine learning workflows to enable rapid tissue identification during surgical procedures.
The resulting platform supports real-time clinical decision making by allowing clinicians to distinguish between normal and diseased tissue directly in the operating suite.
Challenge
The system required integration of advanced optical imaging technology capable of performing rapid biochemical analysis during surgery. Reliable performance was required across demanding clinical environments and complex workflows.
Delivering this capability required precise integration of optical, mechanical, software, and machine learning systems into a clinically deployable architecture.
Client
BlackLight Surgical
Core Disciplines
Solution
Triple Ring assembled multidisciplinary engineering and scientific teams to design and integrate the imaging platform using structured development and validation methodologies.
Technical execution focused on:
- Integrating laser-based optical imaging technologies capable of rapid tissue analysis
- Developing machine learning workflows that supported real-time tissue classification
- Using simulation and modeling tools to guide system design and performance optimization
- Establishing ISO 13485-compliant design and documentation processes
Outcome
Triple Ring delivered a fully integrated intra-operative biochemical imaging platform supporting clinical studies and real-time tissue visualization.
The system was developed to ISO 13485 standards and documented within a Quality Management System transferred to BlackLight Surgical, enabling deployment of advanced intra-operative imaging workflows.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined high-speed laser optical imaging, machine learning, mechanical engineering, and quality systems expertise to develop a fully integrated intra-operative tissue analysis platform capable of distinguishing healthy from diseased tissue in real time.
Todd, Cameran, and Shehadeh collaborated with many talented colleagues across Triple Ring and BlackLight Surgical on this project.

Todd Harris, PhD
Physics & Optical Science
Dr. Todd Harris applies expertise in optics and imaging physics to the development of advanced sensing and illumination technologies. His work combines optical modeling with system design, helping teams translate complex physical principles into dependable, high-performance solutions.

Cameran Casale
Bioengineering
Cameran Casale contributes to the development of imaging, microfluidic, and diagnostic technologies across multidisciplinary programs. Her work supports system integration and testing efforts, helping teams refine complex devices for reliable performance in research and clinical environments.

Shehadeh Dajani
Aerospace & Mechanical Engineering
Shehadeh Dajani supports the development of safety-critical embedded systems used in regulated medical technologies. His work spans software and system integration across feasibility, clinical, and production stages, helping ensure reliable performance throughout the development lifecycle.

Veterinary Vital Sign Monitor
Client
One Health Group
Practice Areas
Core Disciplines
Overview
One Health Group partnered with Triple Ring to develop a non-invasive wearable physiological monitoring system designed to capture biometric data from animals in real time. The system was developed to support continuous health monitoring in veterinary settings while improving patient comfort and enabling early detection of health issues.
The resulting wearable platform integrates physiological sensing and wireless communication technologies to enable long-term monitoring and provide actionable health insights for veterinary care providers.
Challenge
One Health Group required development of a wearable monitoring device capable of accurately measuring physiological signals in animals while remaining comfortable and suitable for continuous use. The system needed to support long-term data collection and deliver real-time alerts without interfering with normal animal movement.
As a lean startup organization, One Health Group relied on a fully outsourced R&D model. The project required multidisciplinary engineering expertise to design, prototype, and validate a complex sensing platform while rapidly demonstrating feasibility and reducing development risk.
Veterinary Vital Sign Monitor
Overview
One Health Group partnered with Triple Ring to develop a non-invasive wearable physiological monitoring system designed to capture biometric data from animals in real time. The system was developed to support continuous health monitoring in veterinary settings while improving patient comfort and enabling early detection of health issues.
The resulting wearable platform integrates physiological sensing and wireless communication technologies to enable long-term monitoring and provide actionable health insights for veterinary care providers.
Challenge
One Health Group required development of a wearable monitoring device capable of accurately measuring physiological signals in animals while remaining comfortable and suitable for continuous use. The system needed to support long-term data collection and deliver real-time alerts without interfering with normal animal movement.
As a lean startup organization, One Health Group relied on a fully outsourced R&D model. The project required multidisciplinary engineering expertise to design, prototype, and validate a complex sensing platform while rapidly demonstrating feasibility and reducing development risk.
Client
One Health Group
Practice Areas
Core Disciplines

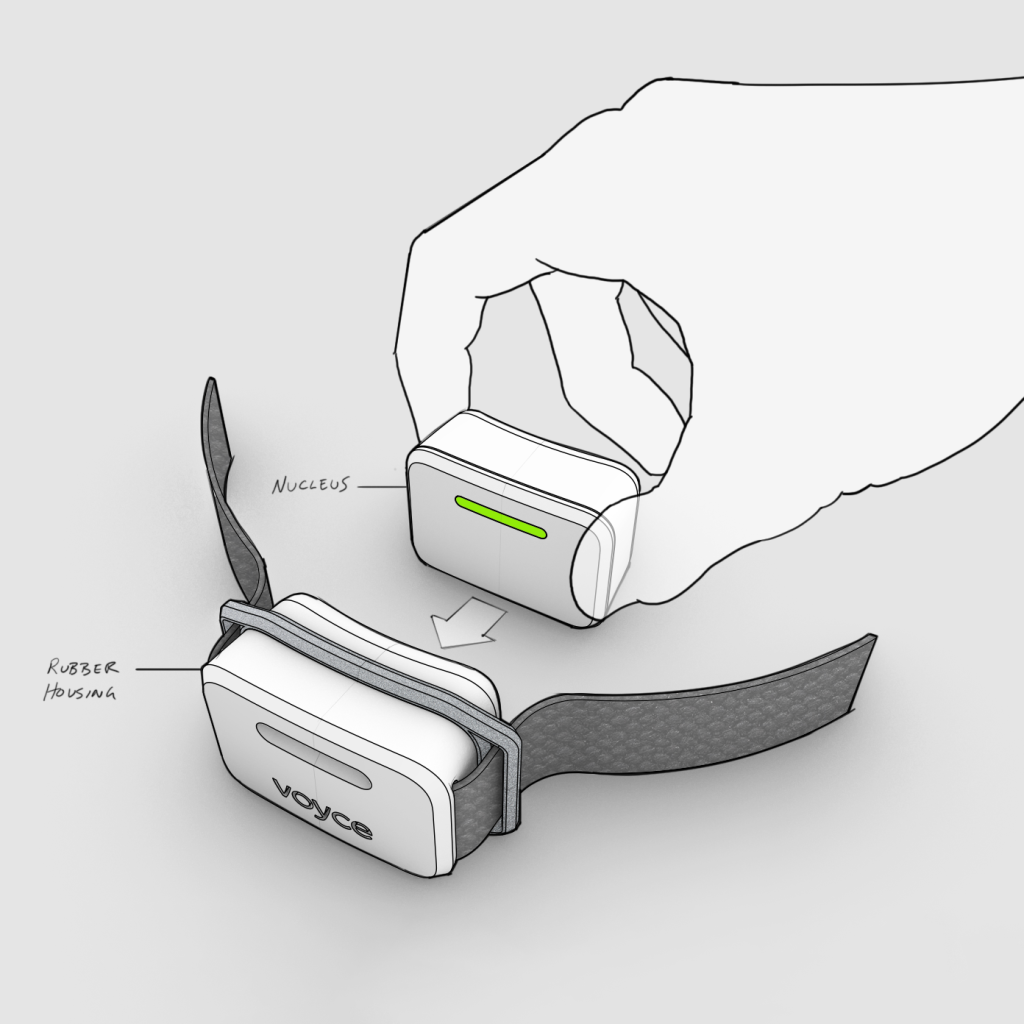

Solution
Triple Ring led the development of a wearable physiological monitoring platform using a structured feasibility and product development approach. Industrial design, sensing technologies, and system integration were coordinated to ensure reliable performance and comfort across extended wear conditions.
Key contributions included:
- Designing wearable hardware capable of supporting continuous physiological monitoring
- Conducting rapid feasibility testing to validate sensing and alert functionality
- Refining industrial design to improve comfort, usability, and durability

Outcome
Triple Ring delivered robust functional prototypes capable of generating high-quality physiological data suitable for ongoing product development and validation. The system demonstrated the feasibility of continuous biometric monitoring in veterinary applications.
The successful development effort supported One Health Group’s technology roadmap and strengthened its value proposition, enabling expanded partnerships with strategic investors in the veterinary health industry.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined physiological sensing, wireless systems, industrial design, and startup-focused R&D expertise to develop a wearable continuous monitoring platform capable of capturing real-time biometric data from animals in veterinary settings.
Brian and Steve collaborated with many talented colleagues across Triple Ring and One Health Group on this project.

Brian Wilfley, PhD
Experimental Physics & Instrumentation
Dr. Brian Wilfley leads the development of complex measurement and instrumentation systems grounded in experimental physics. His work combines theoretical insight with hands-on experimentation to advance technologies used in imaging, inspection, and scientific analysis.

Steve Kuhn
Embedded Systems & Software Engineering
Steve Kuhn develops embedded software and real-time control systems that support complex hardware-driven technologies. His work focuses on software architecture, motion control, and system simulation, enabling reliable operation across integrated device platforms.
Microneedle Patch Applicator
Client
Confidential
Practice Areas
Core Disciplines
Overview
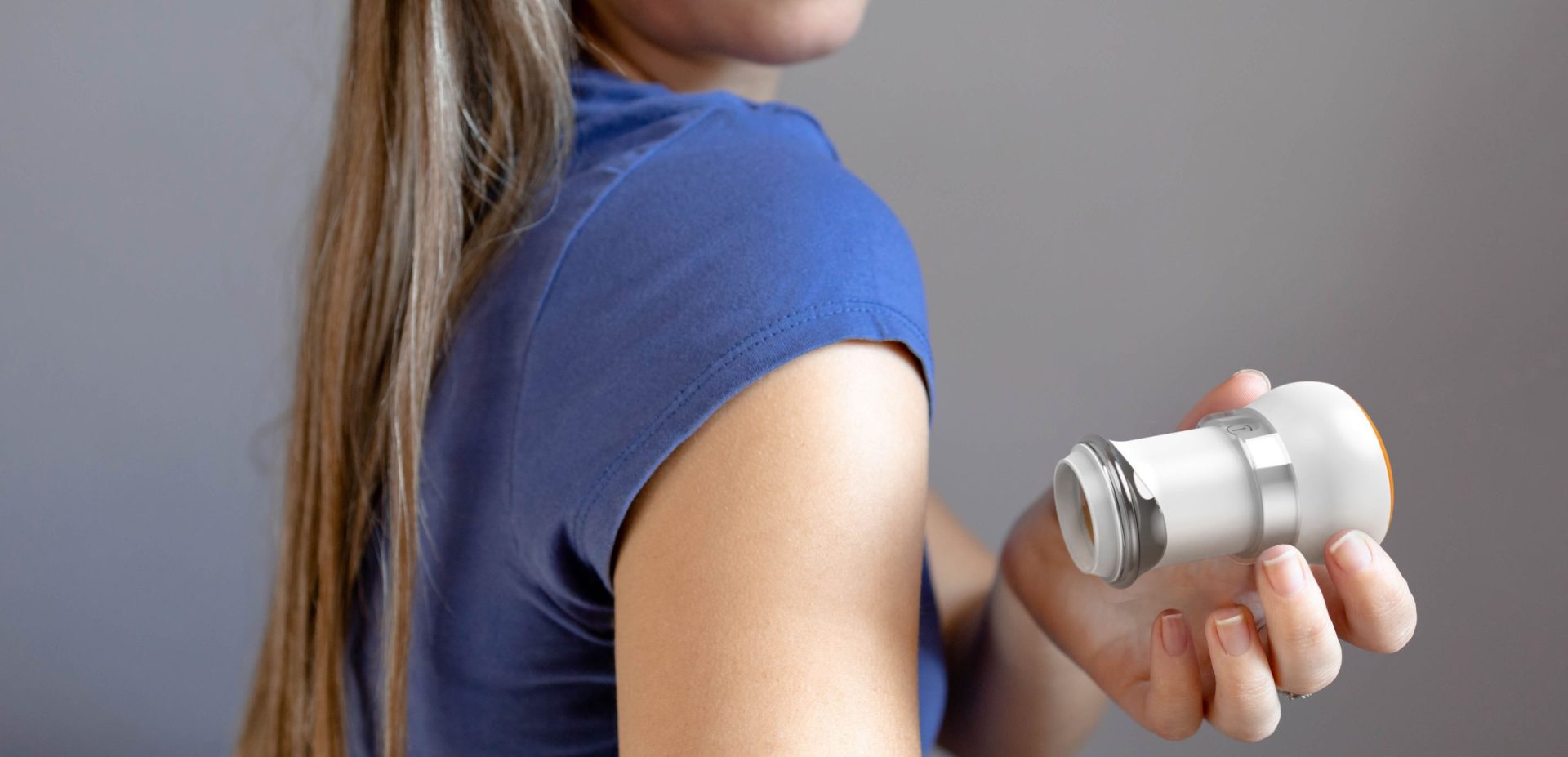
The client selected Triple Ring to design a reusable applicator supporting the delivery of a novel micro-needle patch for transdermal drug administration. The system was intended for patient-administered use in home settings and required consistent mechanical performance to ensure reliable drug delivery.
The resulting combination product integrated a reusable applicator with a micro-needle patch system designed to enable uniform pressure application, supporting consistent adhesion and controlled dosing across diverse patient populations.
Challenge
The client developed a microneedle-based transdermal patch that required significantly greater uniformity of application pressure compared to traditional adhesive patches. Achieving reliable drug delivery required development of a reusable applicator capable of delivering consistent mechanical force during use.
The applicator needed to function effectively across a wide range of skin types, including variations in thickness, age, moisture content, and anatomical placement. In addition to performance requirements, the design needed to meet durability expectations for repeated home use while maintaining low production cost.
Microneedle Patch Applicator
Overview
The client selected Triple Ring to design a reusable applicator supporting the delivery of a novel micro-needle patch for transdermal drug administration. The system was intended for patient-administered use in home settings and required consistent mechanical performance to ensure reliable drug delivery.
The resulting combination product integrated a reusable applicator with a micro-needle patch system designed to enable uniform pressure application, supporting consistent adhesion and controlled dosing across diverse patient populations.
Challenge
The client developed a microneedle-based transdermal patch that required significantly greater uniformity of application pressure compared to traditional adhesive patches. Achieving reliable drug delivery required development of a reusable applicator capable of delivering consistent mechanical force during use.
The applicator needed to function effectively across a wide range of skin types, including variations in thickness, age, moisture content, and anatomical placement. In addition to performance requirements, the design needed to meet durability expectations for repeated home use while maintaining low production cost.
Client
Confidential
Practice Areas
Core Disciplines

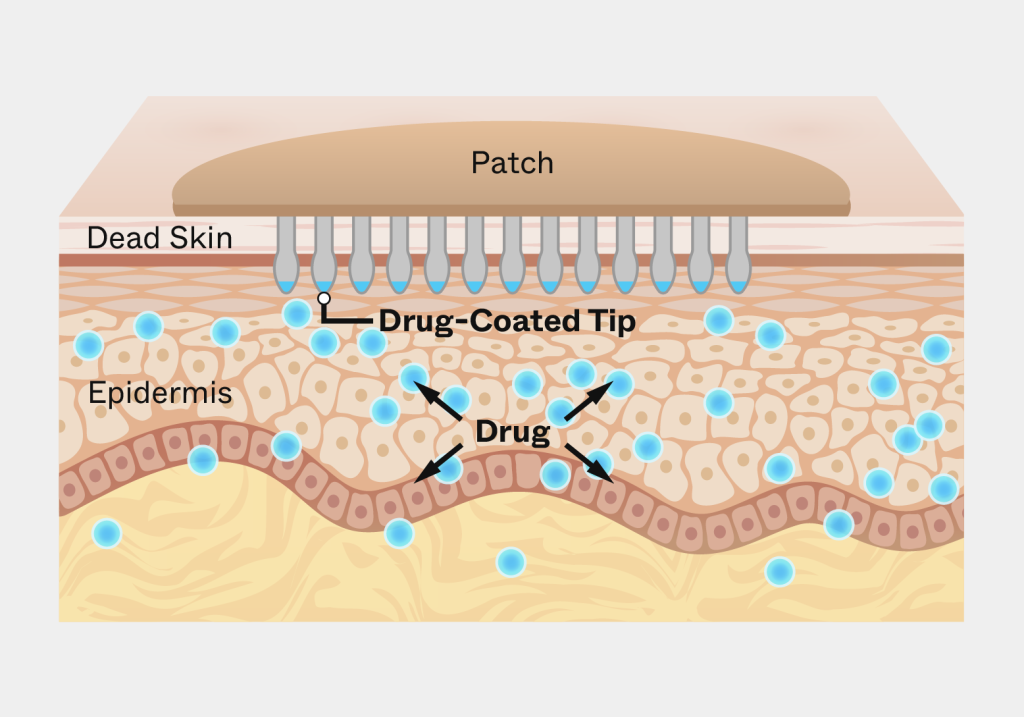
Solution
Triple Ring applied interdisciplinary expertise across materials science, mechanical engineering, and life sciences to design an applicator capable of delivering controlled pressure during patch placement. The development effort focused on optimizing performance across diverse user conditions while maintaining manufacturability and reliability.
Key development efforts included:
- Designing precision mechanical systems capable of delivering uniform pressure during patch application
- Selecting materials optimized for durability, manufacturability, and patient-safe use
- Engineering applicator geometry to accommodate variation in skin thickness, moisture, and elasticity
- Validating device performance across simulated patient-use conditions
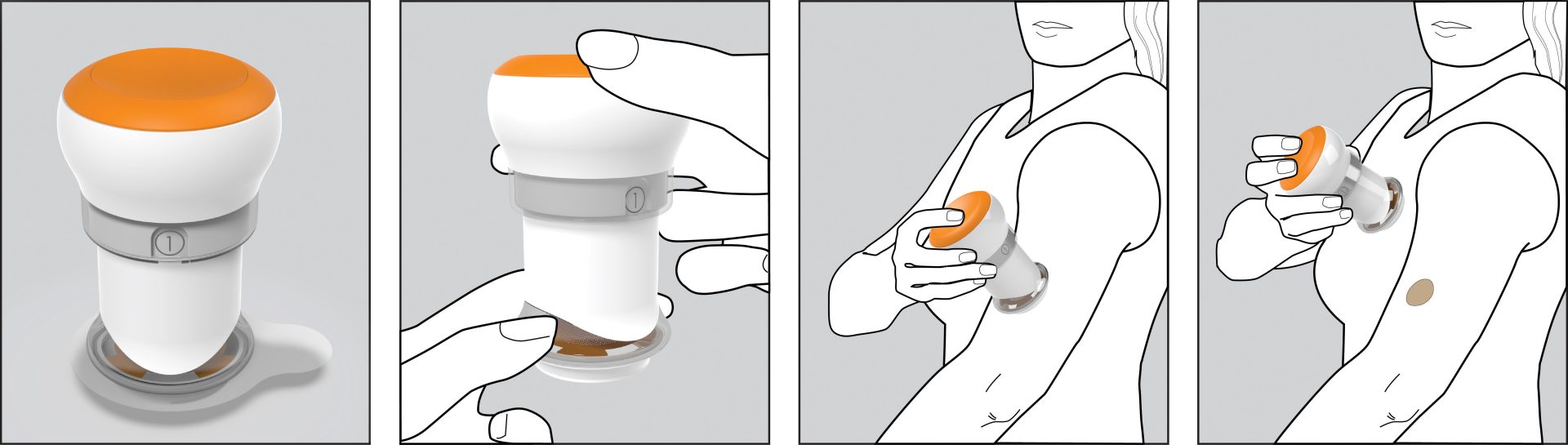
Outcome
Triple Ring delivered a reusable applicator integrated with a micro-needle array patch, forming a single-entity combination product designed for intracutaneous drug delivery. The system enabled rapid drug absorption while supporting consistent dosing performance.
The completed applicator enabled the client to advance into clinical trials and demonstrate the effectiveness of the technology to healthcare stakeholders. The underlying technology was ultimately acquired by a vaccine manufacturer, supporting continued development and commercialization.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined mechanical engineering, materials science, and life sciences expertise to design a reusable applicator capable of delivering consistent, uniform pressure across diverse patient skin types — enabling reliable transdermal drug delivery in home settings.
Walt and Gabe collaborated with many talented colleagues across Triple Ring on this project.

Walt Cecka
Medical Device Design & Translational Medicine
Walt Cecka brings decades of experience translating novel medical technologies into first-to-market products. He works closely with innovators to shape early concepts into structured development programs that advance patient care across a wide range of clinical applications.

Gabriel Chow, PhD
Materials Characterization & Development
Dr. Gabe Chow specializes in materials characterization and mechanical system development across macro to nanoscale environments. His work supports the design, testing, and reliability of advanced materials and mechanical systems, helping translate innovative concepts into durable, real-world solutions.


Kuri, the Nimble Companion Robot
Client
Mayfield Robotics
Practice Areas
Core Disciplines
Overview
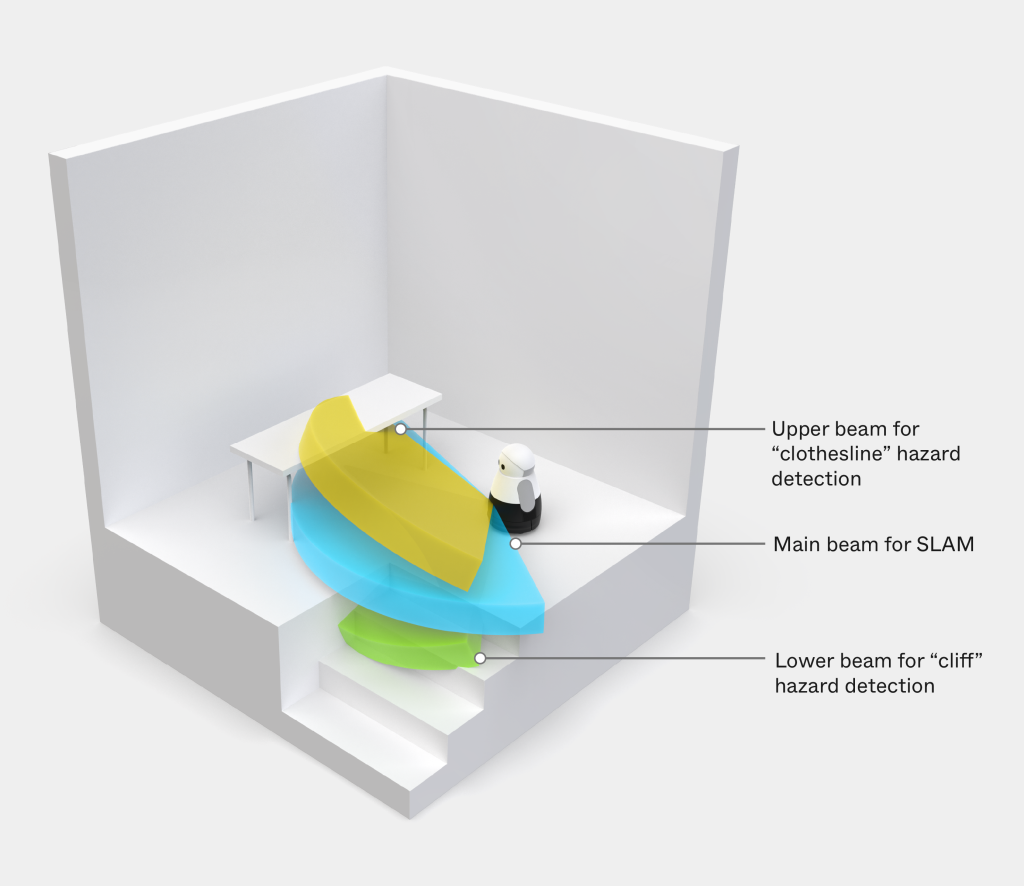
Mayfield Robotics collaborated with Triple Ring to design and develop an optical guidance system for Kuri, a home companion robot designed to safely navigate and interact within household environments. The system required advanced sensing capabilities to support navigation, hazard detection, and environmental awareness.
The resulting optical platform integrated multiple sensing technologies into a compact architecture capable of supporting simultaneous localization and mapping (SLAM) while meeting strict safety and cost requirements for consumer deployment.
Challenge
Kuri required a sophisticated optical system capable of performing multiple functions, including hazard detection, navigation, and real-time environmental mapping. The system needed to maintain high performance in dynamic home environments while remaining safe for use around humans and pets.
Additionally, the integrated optics needed to meet strict eye-safety certification requirements and support reliable operation under consumer use conditions. Designing this system required combining complex optical components within a compact and manufacturable architecture while meeting aggressive launch timelines.
Kuri, the Nimble Companion Robot
Overview
Mayfield Robotics collaborated with Triple Ring to design and develop an optical guidance system for Kuri, a home companion robot designed to safely navigate and interact within household environments. The system required advanced sensing capabilities to support navigation, hazard detection, and environmental awareness.
The resulting optical platform integrated multiple sensing technologies into a compact architecture capable of supporting simultaneous localization and mapping (SLAM) while meeting strict safety and cost requirements for consumer deployment.
Challenge
Kuri required a sophisticated optical system capable of performing multiple functions, including hazard detection, navigation, and real-time environmental mapping. The system needed to maintain high performance in dynamic home environments while remaining safe for use around humans and pets.
Additionally, the integrated optics needed to meet strict eye-safety certification requirements and support reliable operation under consumer use conditions. Designing this system required combining complex optical components within a compact and manufacturable architecture while meeting aggressive launch timelines.
Client
Mayfield Robotics
Practice Areas
Core Disciplines

Solution
Triple Ring applied deep expertise in optical system design and robotics integration to develop a navigation system capable of supporting safe and reliable robot operation. The development effort focused on integrating advanced sensing technologies into a scalable and cost-effective architecture.
Engineering efforts included:
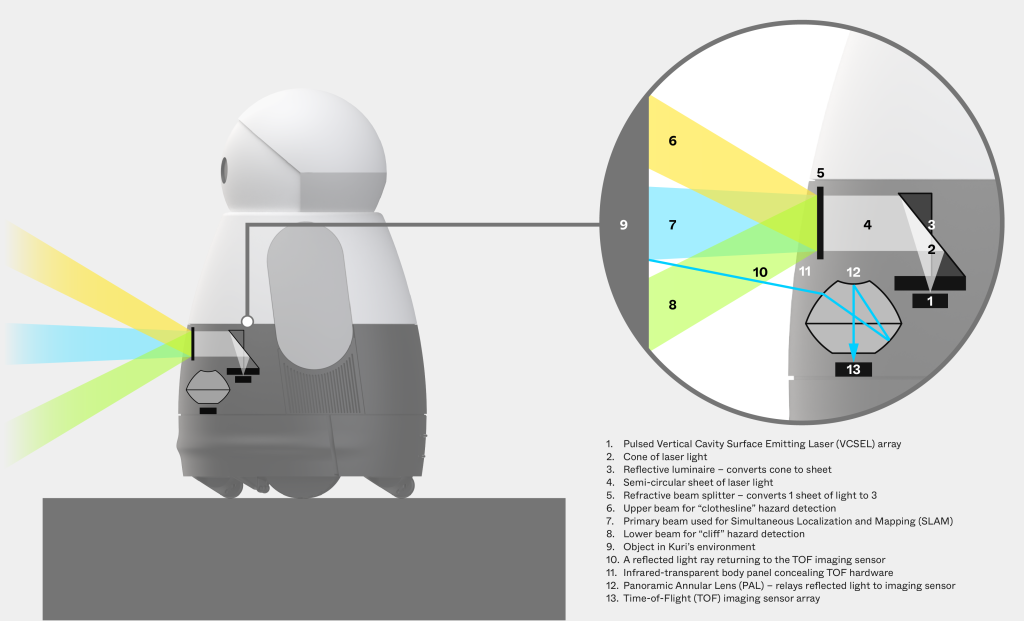
- Designing pulsed vertical cavity surface emitting laser (VCSEL) array systems supporting depth sensing
- Developing panoramic annular lens configurations enabling wide-area environmental awareness
- Integrating time-of-flight imaging sensor arrays for real-time distance measurement
- Engineering optical subsystems compliant with stringent eye-safety requirements
- Supporting system-level integration and validation to meet cost, performance, and production goals

Outcome
Triple Ring delivered an optical navigation system that exceeded cost and performance requirements while achieving certification for eye safety and compliance with UL 3300 standards. The system enabled reliable navigation and safe operation within home environments.
The completed optical system supported the successful commercial launch of Kuri, with hundreds of units delivered to customers. The design generated multiple patent filings and contributed to Kuri being named Best in Show at CES 2017 by leading technology publications.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined optical system design, VCSEL and time-of-flight sensing, robotics integration, and consumer safety certification expertise to develop a compact, eye-safe optical navigation system capable of supporting simultaneous localization, mapping, and hazard detection in dynamic home environments.
Todd collaborated with many talented colleagues across Triple Ring and Mayfield Robotics on this project.

Todd Harris, PhD
Physics & Optical Science
Dr. Todd Harris applies expertise in optics and imaging physics to the development of advanced sensing and illumination technologies. His work combines optical modeling with system design, helping teams translate complex physical principles into dependable, high-performance solutions.

IVD Platform Refresh
Client
Fortune 500 IVD Company
Practice Areas
Core Disciplines
Overview
A Fortune 500 diagnostics manufacturer worked with Triple Ring to modernize a legacy in vitro diagnostic (IVD) platform facing component obsolescence and software limitations. The project focused on refreshing the system architecture while maintaining regulatory equivalency with an existing FDA-cleared product.
The resulting platform replaced obsolete hardware, migrated legacy software, and introduced updated industrial design elements while preserving compatibility with established manufacturing and regulatory pathways.
Challenge
The client faced the obsolescence of critical hardware components, including single-board computers and microcontrollers, within an existing diagnostic platform. Compounding the challenge, original firmware source code was unavailable, and institutional knowledge associated with the system had diminished over time.
In addition to restoring functionality, the refreshed system needed to maintain regulatory equivalence to the original device in order to qualify for a Special 510(k) submission. This requirement demanded careful reverse engineering, system validation, and modernization without introducing unintended performance deviations.
IVD Platform Refresh
Overview
A Fortune 500 diagnostics manufacturer worked with Triple Ring to modernize a legacy in vitro diagnostic (IVD) platform facing component obsolescence and software limitations. The project focused on refreshing the system architecture while maintaining regulatory equivalency with an existing FDA-cleared product.
The resulting platform replaced obsolete hardware, migrated legacy software, and introduced updated industrial design elements while preserving compatibility with established manufacturing and regulatory pathways.
Challenge
The client faced the obsolescence of critical hardware components, including single-board computers and microcontrollers, within an existing diagnostic platform. Compounding the challenge, original firmware source code was unavailable, and institutional knowledge associated with the system had diminished over time.
In addition to restoring functionality, the refreshed system needed to maintain regulatory equivalence to the original device in order to qualify for a Special 510(k) submission. This requirement demanded careful reverse engineering, system validation, and modernization without introducing unintended performance deviations.
Client
Fortune 500 IVD Company
Practice Areas
Core Disciplines


Solution
Triple Ring implemented a structured modernization strategy combining reverse engineering, hardware redesign, and software migration. The development effort focused on preserving functional equivalency while introducing modern components and improving long-term maintainability.
Development efforts included:
- Reverse-engineering legacy hardware and firmware to recover system functionality and requirements
- Migrating existing software to a modern operating system architecture
- Replacing obsolete components while maintaining compatibility with legacy subsystems
- Updating mechanical and electrical subassemblies to support manufacturing continuity
- Preparing validation and documentation packages supporting regulatory equivalence requirements
Outcome
Triple Ring delivered a fully refreshed IVD platform supported by a comprehensive regulatory submission package demonstrating equivalency to the original FDA-cleared device. The updated system maintained functional continuity while addressing long-term hardware and software sustainability.
The refreshed design integrated seamlessly into existing manufacturing workflows, enabling uninterrupted production and maintaining cost targets. The modernization effort positioned the platform for continued market competitiveness while ensuring regulatory compliance and operational reliability.
Triple Ring Talent
The Story Behind the Innovation
At Triple Ring, we draw on a deep bench of expertise across diverse disciplines matched to each innovation challenge. For this project, our team combined reverse engineering, embedded systems, software migration, regulatory strategy, and industrial design expertise to modernize a legacy IVD platform — restoring long-term sustainability while maintaining full regulatory equivalency with the original FDA-cleared device.
Rob and Anne collaborated with many talented colleagues across Triple Ring on this project.

Robert de Saint Phalle
Interdisciplinary Product Design
Robert de Saint Phalle shapes product design strategies that connect engineering, user experience, and emerging technologies. His work helps teams translate complex ideas into thoughtful, functional products that bring new technologies to life.

Anne Preut
Biomedical & Systems Engineering
Anne Preut leads systems engineering efforts that integrate hardware, firmware, assays, and fluidics across complex diagnostic technologies. Her work aligns cross-functional teams throughout the development lifecycle, helping ensure systems perform reliably from early feasibility through production.